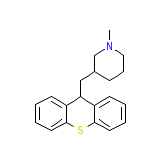
Aneusral




Aneusral Brand names, Aneusral Analogs
- 3P bamate
- Amepromat
- Amosene
- Anastress
- Anathylmon
- Anatimon
- Andaksin
- Andaxin
- Aneural
- Aneurol
- Aneusral
- Aneuxal
- Aneuxral
- Ansiatan
- Ansietan
- Ansil
- Ansiowas
- Anural
- Anxietil
- Anzil
- Apascil
- Apasil
- Apo-Meprobamate
- Appetrol
- Appetrol-Sr
- Arcoban
- Arpon
- Artolon
- Ataraxine
- Atraxin
- Auxietil
- Ayeramate
- Ayermate
- Bamate
- Bamd 400
- Bamo 400
- Biobamat
- Biobamate
- Brobamate
- Calmadin
- Calmax
- Calmiren
- Canquil 400
- Canquil-400
- Carbaxin
- Cirpon
- Cirponyl
- Coprobate
- Crestanil
- Cypron
- Cyrpon
- DEA No. 2820
- Dapaz
- Deprol
- Despasmol
- Dicandiol
- Diron
- Diurnal
- Diurnaldiverondormabrol
- Diveron
- Dormabrol
- Ecuanil
- Edenal
- Enorden
- Epicur
- Epikur
- Equagesic
- Equanil
- Equanil suspension
- Equatrate
- Equazine-M
- Equilium
- Equinil
- Equitar
- Erina
- Estasil
- Fas-Cile
- Fas-Cile 200
- Gadexyl
- Gagexyl
- Harmonin
- Hartol
- Holbamate
- Ipsotian
- Kesso-Bamate
- Kessobamate
- Klort
- Larten
- Lepenil
- Lepetown
- Letyl
- Libiolan
- Madiol
- Mar-Bate
- Margonil
- Mendel
- Mepamtin
- Mepantin
- Mepavlon
- Mepiosine
- Meposed
- Mepranil
- Mepriam
- Meprin
- Meprindon
- Mepro-Aspirin
- Mepro-analgesic
- Meprobam
- Meprobamat
- Meprobamat [German]
- Meprobamate [USAN:BAN:INN:JAN]
- Meprobamate and Aspirin Tablets
- Meprobamato
- Meprobamato [INN-Spanish]
- Meprobamato [Italian]
- Meprobamatum [INN-Latin]
- Meproban
- Meprocompren
- Meprocon
- Meprocon CMC
- Meprodil
- Meprodiol
- Meprol
- Meproleaf
- Mepron
- Mepronil
- Meprosa
- Meprosan
- Meprosin
- Meprospan
- Meprospan 200
- Meprospan 400
- Meprospan-200
- Meprospan-400
- Meprotabs
- Meprotan
- Meprotanum
- Meproten
- Meprotil
- Meprovan
- Meprovanmeprozine
- Meprozine
- Meptran
- Meptranactylmilprem
- Mesmar
- Metractyl
- Metranquil
- Micrainin
- Milpath
- Milprem
- Milprem-200
- Milprem-400
- Miltamato
- Miltann
- Miltaun
- Miltown
- Miltown 600
- Miltown-200
- Miltown-400
- Miltown-600
- Miltrate
- Miltuan
- Miltwon
- Misedant
- Morbam
- Multaun
- My-trans
- Neo-Tran
- Nephentine
- Nervonus
- Neuramate
- Oasil
- Optarket
- Orlevol
- Orolevol
- Pan-tranquil
- Pancalma
- Panediol
- Pankalma
- Pathibamate
- Paxin
- Pensive
- Perequietil
- Perequil
- Perquietil
- Pertranquil
- Pimal
- Placidon
- Placitate
- Prequil
- Probamato
- Probamyl
- Probate
- Procalmadiol
- Procalmadol
- Procalmidol
- Procarbamide
- Promate
- Promato
- Proquanil
- Protran
- Q-Gesic
- Quaname
- Quanane
- Quanil
- Quietidon
- Quivet
- Rastenil
- Reostral
- Restenil
- Restenyl
- Restinal
- Restinil
- Restran
- Robamate
- SK-Bamate
- Sadanyl
- Scolazil
- Sedabamate
- Sedanil
- Sedanyl
- Sedazil
- Sedoquil
- Sedoselecta
- Selene
- Seril
- Setran
- Shalvaton
- Solevione anastress
- Sowell
- Spantran
- Stensolo
- Tamate
- Tensol
- Tensonal
- Trancot
- Trankvilan
- Tranlisant
- Tranmep
- Tranquil
- Tranquilan
- Tranquilate
- Tranquilax
- Tranquiline
- Tranquillin
- Tranquilsan
- Tranquinol
- Tranquisan
- Trelmar
- Urbil
- Urbilat
- Vio-Bamate
- Vistabamate
- Wardamate
- Wyseals
- Zirpon
Aneusral Brand Names Mixture
- No information avaliable
Aneusral Chemical_Formula
C20H23NS
Aneusral RX_link
No information avaliable
Aneusral fda sheet
Aneusral msds (material safety sheet)
Aneusral Synthesis Reference
No information avaliable
Aneusral Molecular Weight
309.469 g/mol
Aneusral Melting Point
< 25 oC
Aneusral H2O Solubility
Soluble as HCl salt
Aneusral State
Liquid
Aneusral LogP
5.7
Aneusral Dosage Forms
Tablets (oral, 1 mg)
Aneusral Indication
Used for the symptomatic treatment of parkinsonism.
Aneusral Pharmacology
Metixene is a tertiary antimuscarinic with actions similar to those of atropine; it also has antihistaminic and direct antispasmodic properties. It is used for the symptomatic treatment of parkinsonism, including the alleviation of the extrapyramidal syndrome induced by other drugs such as phenothiazines, but, like other antimuscarinics, it is of no value against tardive dyskinesias. Metixene has been discontinued.
Aneusral Absorption
Absorbed in the gastrointestinal tract following oral administration, however the extent of absorption is not known.
Aneusral side effects and Toxicity
Signs of overdose include dilated and sluggish pupils, warm, dry skin, facial flushing, decreased secretions of the mouth, pharynx, nose, and bronchi, foul-smelling breath, elevated temperature, tachycardia, cardiac arrhythmias, decreased bowel sounds, urinary retention, delirium, disorientation, anxiety, hallucinations, illusions, confusion, incoherence, agitation, hyperactivity, ataxia, loss of memory, paranoia, combativeness, and seizures.
Aneusral Patient Information
Aneusral Organisms Affected
Humans and other mammals