Duranifin




Duranifin Brand names, Duranifin Analogs
- Adalat
- Adalat 10
- Adalat 20
- Adalat 5
- Adalat CC
- Adalat CR
- Adalat Crono
- Adalat Ft
- Adalat Gits
- Adalat Gits 30
- Adalat LA
- Adalat LP
- Adalat Oros
- Adalat PA
- Adalat Retard
- Adalate
- Adapine
- Adapress
- Alat
- Aldipin
- Alfadal
- Alonix
- Alonix S
- Alpha-Nifedipine Retard
- Angipec
- Anifed
- Anpine
- Apo-Nifed
- Aprical
- Bonacid
- Calcibloc
- Calcigard
- Calcilat
- Camont
- Cardifen
- Cardilat
- Cardilate
- Cardionorm
- Chronadalate
- Chronadalate Lp
- Citilat
- Coracten
- Coral
- Cordafen
- Cordaflex
- Cordalat
- Cordicant
- Cordilan
- Cordipin
- Corinfar
- Corotrend
- Corynphar
- Depin
- Dignokonstant
- Dilafed
- Dilcor
- Dipinkor
- Duranifin
- Ecodipi
- Ecodipin
- Ecodipin E
- Fedcor
- Fedcor Retard
- Fenamon
- Fenamon Sr
- Fenihidin
- Fenihidine
- Glopir
- Hadipin
- Hexadilat
- Introcar
- Kordafen
- Macorel
- Megalat
- Myogard
- N1fedilat
- Nedipin
- Nicardia
- Nifangin
- Nifar
- Nifdemin
- Nifebene
- Nifecard
- Nifecor
- Nifedepat
- Nifedicor
- Nifedin
- Nifedine
- Nifedipine Retard
- Nifedipres
- Nifedirex LP
- Nifelan
- Nifelat
- Nifelat Q
- Nifelate
- Nifensar XL
- Nificard
- Nifidine
- Nifipen
- Niphedipine
- Orix
- Oxcord
- Pidilat
- Procardia
- Procardia XL
- Sepamit
- Tibricol
- Zenusin
Duranifin Brand Names Mixture
- No information avaliable
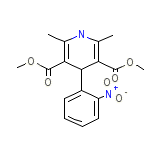
Duranifin Chemical_Formula
C17H18N2O6
Duranifin RX_link
http://www.rxlist.com/cgi/generic2/nifedip.htm
Duranifin fda sheet
Duranifin msds (material safety sheet)
Duranifin Synthesis Reference
Bossert, Vater, U.S. Pat. 3,485,847 (1969)
Duranifin Molecular Weight
346.335 g/mol
Duranifin Melting Point
172 - 174 oC
Duranifin H2O Solubility
Insoluble
Duranifin State
Solid
Duranifin LogP
2.343
Duranifin Dosage Forms
Capsule; Tablet; Tablet (extended-release)
Duranifin Indication
For the management of vasospastic angina, chronic stable angina and hypertension.
Duranifin Pharmacology
Nifedipine, the prototype of the dihydropyridine class of calcium-channel antagonists, is similar to other dihydropyridines including amlodipine, felodipine, isradipine, and nicardipine. Nifedipine is used to treat Prinzmetal's angina, hypertension, and other vascular disorders such as Raynaud's phenomenon. By blocking the calcium channels, Nifedipine inhibits the spasm of the coronary artery and dilates the systemic arteries, results in a increase of myocardial oxygen supply and a decrease in systemic blood pressure.
Duranifin Absorption
Rapidly and fully absorbed following oral administration.
Duranifin side effects and Toxicity
Symptoms of overdose include dizziness, drowsiness, nausea, severe drop in blood pressure, slurred speech, and weakness. LD50=494 mg/kg (orally in mice); LD50=1022 mg/kg (orally in rats)
Duranifin Patient Information
Duranifin Organisms Affected
Humans and other mammals