Condition




Condition Brand names, Condition Analogs
- Alboral
- Aliseum
- Alupram
- Amiprol
- An-Ding
- Ansiolin
- Ansiolisina
- Apaurin
- Apo-Diazepam
- Apozepam
- Armonil
- Assival
- Atensine
- Atilen
- Bensedin
- Bialzepam
- Calmocitene
- Calmpose
- Cercine
- Ceregulart
- Condition
- DAP
- Diacepan
- Dialag
- Dialar
- Diapam
- Diastat
- Diazemuls
- Diazemulus
- Diazepam Intensol
- Diazepan
- Diazetard
- Dienpax
- Dipam
- Dipezona
- Dizac
- Domalium
- Duksen
- Duxen
- E-Pam
- Eridan
- Eurosan
- Evacalm
- Faustan
- Faustan,
- Freudal
- Frustan
- Gewacalm
- Gihitan
- Horizon
- Kabivitrum
- Kiatrium
- LA III
- La-Iii
- Lamra
- Lembrol
- Levium
- Liberetas
- Mandrozep
- Methyldiazepinone
- Methyldiazepinone, Pharmaceutical
- Morosan
- Neurolytril
- Noan
- Novazam
- Novo-Dipam
- Paceum
- Pacitran
- Paranten
- Paxate
- Paxel
- Plidan
- Pms-Diazepam
- Pro-Pam
- Q-Pam
- Q-Pam Relanium
- Quetinil
- Quiatril
- Quievita
- Relaminal
- Relanium
- Relax
- Renborin
- Ruhsitus
- S.A. R.L.
- Saromet
- Sedapam
- Sedipam
- Seduksen
- Seduxen
- Serenack
- Serenamin
- Serenzin
- Servizepam
- Setonil
- Sibazon
- Sibazone
- Solis
- Sonacon
- Stesolid
- Stesolin
- Tensopam
- Tranimul
- Tranqdyn
- Tranquase
- Tranquirit
- Tranquo-Puren
- Tranquo-Tablinen
- Umbrium
- Unisedil
- Usempax Ap
- Valaxona
- Valeo
- Valiquid
- Valitran
- Valium
- Valrelease
- Vatran
- Velium
- Vival
- Vivol
- Zetran
- Zipan
Condition Brand Names Mixture
- No information avaliable
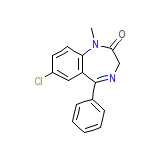
Condition Chemical_Formula
C16H13ClN2O
Condition RX_link
http://www.rxlist.com/cgi/generic/diazepam.htm
Condition fda sheet
Condition msds (material safety sheet)
Condition Synthesis Reference
Reeder, Sternbach; U.S. Pat. 3,371,085 (1968)
Condition Molecular Weight
284.74 g/mol
Condition Melting Point
125-126oC
Condition H2O Solubility
Slightly soluble (50 mg/L)
Condition State
solid
Condition LogP
2.988
Condition Dosage Forms
Tablets; Injectable solution
Condition Indication
Used in the treatment of severe anxiety disorders, as a hypnotic in the short-term management of insomnia, as a sedative and premedicant, as an anticonvulsant, and in the management of alcohol withdrawal syndrome.
Condition Pharmacology
Diazepam, a benzodiazepine, generates the same active metabolite as chlordiazepoxide and clorazepate. In animals, diazepam appears to act on parts of the limbic system, the thalamus and hypothalamus, and induces calming effects. Diazepam, unlike chlorpromazine and reserpine, has no demonstrable peripheral autonomic blocking action, nor does it produce extrapyramidal side effects; however, animals treated with diazepam do have a transient ataxia at higher doses. Diazepam was found to have transient cardiovascular depressor effects in dogs. Long-term experiments in rats revealed no disturbances of endocrine function. Injections into animals have produced localized irritation of tissue surrounding injection sites and some thickening of veins after intravenous use.
Condition Absorption
Essentially complete, with a bioavailability of 93%.
Condition side effects and Toxicity
Symptoms of overdose include somnolence, confusion, coma, and diminished reflexes. Respiration, pulse and blood pressure should be monitored.
Condition Patient Information
Condition Organisms Affected
Humans and other mammals