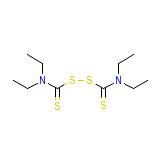
Hocakrotenalnci-C02959




Hocakrotenalnci-C02959 Brand names, Hocakrotenalnci-C02959 Analogs
- Abstensil
- Abstinil
- Abstinyl
- Accel Tet
- Accel Tet-R
- Akrochem Tetd
- Alcophobin
- Alk-Aubs
- Ancazide Et
- Antabus
- Antabuse
- Antadix
- Antaenyl
- Antaethan
- Antaethyl
- Antaetil
- Antalcol
- Antetan
- Antethyl
- Antetil
- Anteyl
- Anthethyl
- Anti-Ethyl
- Antiaethan
- Anticol
- Antietanol
- Antietil
- Antikol
- Antivitium
- Aversan
- Averzan
- Bonibal
- Contralin
- Contrapot
- Cronetal
- Dicupral
- Disetil
- Disulfan
- Disulfram
- Disulfuram
- Disulphuram
- Dupon 4472
- Dupont Fungicide 4472
- Ekagom Dtet
- Ekagom Teds
- Ekagom Tetds
- Ekaland Tetd
- Ephorran
- Espenal
- Esperal
- Etabus
- Ethyl Thiram
- Ethyl Thiudad
- Ethyl Thiurad
- Ethyl Tuads
- Ethyl Tuads Rodform
- Ethyl Tuex
- Ethyldithiourame
- Ethyldithiurame
- Etyl Tuex
- Exhoran
- Exhorran
- Gababentin
- Hoca
- Hocakrotenalnci-C02959
- Krotenal
- Nocbin
- Nocceler Tet
- Nocceler Tet-G
- Noxal
- Perkacit Tetd
- Perkait Tetd
- Refusal
- Ro-Sulfiram
- Sanceler Tet
- Sanceler Tet-G
- Soxinol Tet
- Stopaethyl
- Stopethyl
- Stopety
- Stopetyl
- Super Rodiatox
- TATD
- TETD
- TTD
- TTS
- Tenurid
- Tenutex
- Tetidis
- Tetradin
- Tetradine
- Tetraethylthioperoxydicarbonic Diamide
- Tetraethylthiram Disulfide
- Tetraethylthiram Disulphide
- Tetraethylthiuram
- Tetraethylthiuram Disulfide
- Tetraethylthiuram Disulphide
- Tetraethylthiuram Sulfide
- Tetraethylthiuran Disulfide
- Tetraetil
- Teturam
- Teturamin
- Thiocid
- Thiophos
- Thiosan
- Thioscabin
- Thireranide
- Thiuram Disulfide, Tetraethyl-
- Thiuram E
- Thiuranide
- Tillram
- Tiuram
- Tts X
- Tuads, Ethyl
- Usaf B-33
Hocakrotenalnci-C02959 Brand Names Mixture
- No information avaliable
Hocakrotenalnci-C02959 Chemical_Formula
C10H20N2S4
Hocakrotenalnci-C02959 RX_link
http://www.rxlist.com/cgi/generic/disulfiram.htm
Hocakrotenalnci-C02959 fda sheet
Hocakrotenalnci-C02959 msds (material safety sheet)
Hocakrotenalnci-C02959 Synthesis Reference
No information avaliable
Hocakrotenalnci-C02959 Molecular Weight
296.543 g/mol
Hocakrotenalnci-C02959 Melting Point
71.5 oC
Hocakrotenalnci-C02959 H2O Solubility
4.09 mg/L
Hocakrotenalnci-C02959 State
Solid
Hocakrotenalnci-C02959 LogP
4.422
Hocakrotenalnci-C02959 Dosage Forms
Tablet
Hocakrotenalnci-C02959 Indication
For the treatment and management of chronic alcoholism
Hocakrotenalnci-C02959 Pharmacology
Disulfiram produces a sensitivity to alcohol which results in a highly unpleasant reaction when the patient under treatment ingests even small amounts of alcohol. Disulfiram blocks the oxidation of alcohol at the acetaldehyde stage during alcohol metabolism following disulfiram intake, the concentration of acetaldehyde occurring in the blood may be 5 to 10 times higher than that found during metabolism of the same amount of alcohol alone. Accumulation of acetaldehyde in the blood produces a complex of highly unpleasant symptoms referred to hereinafter as the disulfiram-alcohol reaction. This reaction, which is proportional to the dosage of both disulfiram and alcohol, will persist as long as alcohol is being metabolized. Disulfiram does not appear to influence the rate of alcohol elimination from the body. Prolonged administration of disulfiram does not produce tolerance; the longer a patient remains on therapy, the more exquisitely sensitive he becomes to alcohol.
Hocakrotenalnci-C02959 Absorption
Disulfiram is absorbed slowly from the gastrointestinal tract (80 to 90% of oral dose).
Hocakrotenalnci-C02959 side effects and Toxicity
LD50=8.6g/kg (orally in rats). Symptoms of overdose include irritation, slight drowsiness, unpleasant taste, mild GI disturbances, and orthostatic hypotension.
Hocakrotenalnci-C02959 Patient Information
No information avaliable
Hocakrotenalnci-C02959 Organisms Affected
Humans and other mammals