Mygdalon




Mygdalon Brand names, Mygdalon Analogs
- Apo-Metoclop
- Cerucal
- Clopra
- Clopra-Yellow
- Clopromate
- DEL
- Duraclamid
- Elieten
- Emetid
- Emperal
- Eucil
- Gastrese
- Gastro-Timelets
- Gastrobid
- Gastromax
- Gastronerton
- Gastrosil
- Gastrotablinen
- Gastrotem
- Imperan
- Maxeran
- Maxolon
- Meclopran
- Metaclopramide
- Metaclopromide
- Metamide
- Methochlopramide
- Methoclopramide
- Metochlopramide
- Metoclol
- Metoclopramida [Inn-Spanish]
- Metoclopramide Hcl
- Metoclopramide Hydrochloride
- Metoclopramide Intensol
- Metoclopramide Omega
- Metoclopramidum [Inn-Latin]
- Metocobil
- Metramid
- Moriperan
- Mygdalon
- Neu-Sensamide
- Nu-Metoclopramide
- Octamide
- Parmid
- Paspertin
- Peraprin
- Plasil
- Pms-Metoclopramide
- Pramiel
- Pramin
- Primperan
- Reclomide
- Reglan
- Reliveran
- Terperan
Mygdalon Brand Names Mixture
- No information avaliable
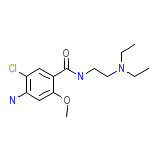
Mygdalon Chemical_Formula
C14H22ClN3O2
Mygdalon RX_link
http://www.rxlist.com/cgi/generic2/metoclo.htm
Mygdalon fda sheet
Mygdalon msds (material safety sheet)
Mygdalon Synthesis Reference
Thominet, U.S. Pat. 3,177,252 (1965)
Mygdalon Molecular Weight
299.796 g/mol
Mygdalon Melting Point
147.25 oC
Mygdalon H2O Solubility
200 mg/L
Mygdalon State
Solid
Mygdalon LogP
2.464
Mygdalon Dosage Forms
Liquid; Syrup; Tablet
Mygdalon Indication
For the treatment of gastroesophageal reflux disease (GERD)
Mygdalon Pharmacology
Metoclopramide, although chemically related to procainamide, does not possess local anesthetic or antiarrhythmic properties. Metoclopramide is used to enhance GI motility, to treat diabetic gastroparesis, as an antinauseant, and to facilitate intubation of the small bowel during radiologic examination. Metoclopramide may be used to treat chemotherapy-induced emesis and as a radiosensitizing agents in the treatment of non-small cell lung carcinoma and glioblastomas in the future.
Mygdalon Absorption
Rapidly and well absorbed (oral bioavailability 80±15.5%).
Mygdalon side effects and Toxicity
Oral, mouse LD50: 280 mg/kg. Signs of overdose include drowsiness, disorientation, and extrapyramidal reactions.
Mygdalon Patient Information
Mygdalon Organisms Affected
Humans and other mammals