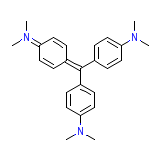
Crystal Violet USP




Crystal Violet USP Brand names, Crystal Violet USP Analogs
- Adergon
- Aizen Crystal Violet
- Aizen Crystal Violet Extra Pure
- Aniline Violet
- Aniline Violet pyoktanine
- Atmonil
- Avermin
- Axuris
- Badil
- Basic Violet 3
- Basic Violet BN
- Basic Violet-3
- Bismuth Violet
- Brilliant Violet 5B
- C.I. Basic Violet 3
- Calcozine Violet 6BN
- Calcozine Violet C
- Crystal Violet
- Crystal Violet 10B
- Crystal Violet 5BO
- Crystal Violet 6B
- Crystal Violet 6BO
- Crystal Violet AO
- Crystal Violet AON
- Crystal Violet BP
- Crystal Violet BPC
- Crystal Violet Extra Pure
- Crystal Violet Extra Pure APN
- Crystal Violet Extra Pure APNX
- Crystal Violet FN
- Crystal Violet HL2
- Crystal Violet O
- Crystal Violet Pure DSC
- Crystal Violet Pure DSC Brilliant
- Crystal Violet SS
- Crystal Violet Technical
- Crystal Violet USP
- Crystal Violet base
- Crystal Violet chloride
- Crystal Violet chloride salt
- GV-Eleven
- Genapax
- Gentersal
- Gentiaverm
- Genticid
- Gentioletten
- Gvs
- Hecto Violet R
- Hectograph Violet SR
- Hexamethyl Violet
- Hexamethylpararosaniline chloride
- Hidaco Brilliant Crystal Violet
- Hidaco crystal Violet
- Meroxyl-Wander
- Meroxylan-Wander
- Methyl Violet 10B
- Methyl Violet 10BD
- Methyl Violet 10BK
- Methyl Violet 10BN
- Methyl Violet 10BNS
- Methyl Violet 10BO
- Methyl Violet 5BNO
- Methyl Violet 5BO
- Methyl Violet 6B
- Methyl violet
- Methylrosaniline chloride
- Mitsui Crystal Violet
- Oxiuran
- Oxycolor
- Oxyozyl
- Paper Blue R
- Plastoresin Violet 5BO
- Pyoktanin
- Vermicid
- Vianin
- Viocid
- Violet 5BO
- Violet 6BN
- Violet CP
- Violet XXIII
- crystal violet for microscopy
- gentian violet gr
- gention violet alchoholic staining solution
- methylrosanilinium chloride
Crystal Violet USP Brand Names Mixture
- No information avaliable
Crystal Violet USP Chemical_Formula
C25H30N3+
Crystal Violet USP RX_link
No information avaliable
Crystal Violet USP fda sheet
Crystal Violet USP msds (material safety sheet)
Crystal Violet USP Synthesis Reference
No information avaliable
Crystal Violet USP Molecular Weight
372.526 g/mol
Crystal Violet USP Melting Point
215 oC
Crystal Violet USP H2O Solubility
4 mg/mL
Crystal Violet USP State
Solid
Crystal Violet USP LogP
3.18
Crystal Violet USP Dosage Forms
Liquid; Tincture
Crystal Violet USP Indication
For the treatment of bacterial and fungal infections inside the mouth (thrush) and skin, also for the prevention of transmission of Chagas' disease (as a blood additive).
Crystal Violet USP Pharmacology
Gentian violet is a mutagen, a mitotic poison, and a clastogen. Gentian violet has been used in medicine for almost 100 years: as an antiseptic for external use, as a topical antibiotic, as a topical antifungal agent, as an antihelminthic agent by oral administration, and more recently, as a blood additive to prevent transmission of Chagas' disease. It is thought to work by binding to the DNA of target organisms and causing disruption, mutation or inhibition of DNA replication.
Crystal Violet USP Absorption
No information avaliable
Crystal Violet USP side effects and Toxicity
LD50=420 mg/kg (rat, oral). Oral administration can cause gastrointestinal irritation, and intravenous injection can cause depression in the white blood cell count.
Crystal Violet USP Patient Information
Crystal Violet USP Organisms Affected
Yeast and other fungi, bacteria and protozoans