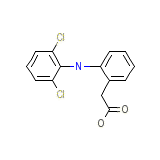
Voltaren-XR




Voltaren-XR Brand names, Voltaren-XR Analogs
- Allvoran
- Apo-Diclo
- Assaren
- Benfofen
- Cataflam
- Delphimix
- Dichlofenac
- Dichronic
- Diclo-Phlogont
- Diclo-Puren
- Diclobenin
- Diclofenac Acid
- Diclofenac Potassium
- Diclofenac Sodium
- Diclord
- Dicloreum
- Dolobasan
- Duravolten
- Ecofenac
- Effekton
- Kriplex
- Neriodin
- Novapirina
- Novo-Difenac
- Novo-Difenac SR
- Nu-Diclo
- Pennsaid
- Primofenac
- Prophenatin
- Rhumalgan
- Solaraze
- Solaraze T
- Tsudohmin
- Valetan
- Voldal
- Voltaren
- Voltaren Ophtha
- Voltaren Ophthalmic
- Voltaren Rapide
- Voltaren SR
- Voltaren-XR
- Voltarol
- Xenid
Voltaren-XR Brand Names Mixture
- No information avaliable
Voltaren-XR Chemical_Formula
C14H11Cl2NO2
Voltaren-XR RX_link
http://www.rxlist.com/cgi/generic/diclofen.htm
Voltaren-XR fda sheet
Voltaren-XR msds (material safety sheet)
Voltaren-XR Synthesis Reference
A. Sallman, R. Pfister, U.S. Pat. 3,558,690, (1971)
Voltaren-XR Molecular Weight
296.148 g/mol
Voltaren-XR Melting Point
283-285oC
Voltaren-XR H2O Solubility
0.00082 mg/mL (Na salt); 2.43 mg/mL (K salt)
Voltaren-XR State
solid
Voltaren-XR LogP
4.218
Voltaren-XR Dosage Forms
Tablet (extended-release); Tablet (immediate-release)
Voltaren-XR Indication
For the acute and chronic treatment of signs and symptoms of osteoarthritis and rheumatoid arthritis.
Voltaren-XR Pharmacology
Diclofenac is an acetic acid nonsteroidal antiinflammatory drug (NSAID) with analgesic and antipyretic properties. Diclofenac is used to treat pain, dysmenorrhea, ocular inflammation, osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, and actinic keratosis
Voltaren-XR Absorption
Completely absorbed from the gastrointestinal tract.
Voltaren-XR side effects and Toxicity
Symptoms of overdose include loss of consciousness, increased intracranial pressure, and aspiration pneumonitis. LD50=390mg/kg (orally in mice)
Voltaren-XR Patient Information
Voltaren-XR Organisms Affected
Humans and other mammals