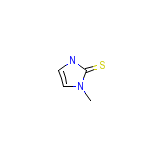
USAF EL-30




USAF EL-30 Brand names, USAF EL-30 Analogs
- Basolan
- Danantizol
- Favistan
- Frentirox
- Mercaptazole
- Mercasolyl
- Mercazole
- Mercazolyl
- Merkastan
- Merkazolil
- Metazolo
- Methamazole
- Methiamazole
- Methimazol
- Methylmercaptoimidazole
- Metizol
- Metothyrin
- Metothyrine
- Metotirin
- Propyl-Thyracil
- Strumazol
- Strumazole
- Tapazole
- Tapuzole
- Thacapzol
- Thiamazol
- Thiamazole
- Thimazole
- Thycapsol
- Thycapzol
- Thymidazol
- Thymidazole
- USAF EL-30
USAF EL-30 Brand Names Mixture
- No information avaliable
USAF EL-30 Chemical_Formula
C4H6N2S
USAF EL-30 RX_link
http://www.rxlist.com/cgi/generic3/methim.htm
USAF EL-30 fda sheet
USAF EL-30 msds (material safety sheet)
USAF EL-30 Synthesis Reference
No information avaliable
USAF EL-30 Molecular Weight
114.17 g/mol
USAF EL-30 Melting Point
146 oC
USAF EL-30 H2O Solubility
275 g/L
USAF EL-30 State
Solid
USAF EL-30 LogP
-0.34
USAF EL-30 Dosage Forms
Tablet (5 or 10 mg)
USAF EL-30 Indication
For the treatment of hyperthyroidism, goiter, Graves disease and psoriasis.
USAF EL-30 Pharmacology
Used in the treatment of hyperthyroidism or an overactive thyroid gland, methimazole inhibits the synthesis of thyroid hormones and thus is effective in the treatment of hyperthyroidism. It may also be used to ameliorate hyperthyroidism in preparation for subtotal thyroidectomy or radioactive iodine therapy.
USAF EL-30 Absorption
Rapid with an oral bioavailability of 93%.
USAF EL-30 side effects and Toxicity
Oral LD50 in rats is 2250 mg/kg. Symptoms of overdose include nausea, vomiting, epigastric distress, headache, fever, joint pain, pruritus, and edema. Aplastic anemia (pancy-topenia) or agranulocytosis may be manifested in hours to days. Less frequent events are hepatitis, nephrotic syndrome, exfoliative dermatitis, neuropathies, and CNS stimulation or depression.
USAF EL-30 Patient Information
Patients should be instructed to report to their physicians any symptoms of agranulocytosis, such as fever or sore throat.
USAF EL-30 Organisms Affected
Humans and other mammals