Predni-Sediv




Predni-Sediv Brand names, Predni-Sediv Analogs
- Algosediv
- Asidon 3
- Asmadion
- Asmaval
- Bonbrain
- Bonbrrin
- Calmore
- Calmorex
- Contergan
- Corronarobetin
- Distaval
- Distaxal
- Distoval
- Ectiluran
- Enterosediv
- Gastrinide
- Glupan
- Glutanon
- Grippex
- Hippuzon
- Imida-Lab
- Imidan
- Imidene
- Isomin
- Kedavon
- Kevadon
- Lulamin
- N-Phthalimidoglutamic acid imide
- N-Phthaloylglutamimide
- N-Phthalylglutamic acid imide
- Neaufatin
- Neo
- Neosedyn
- Neosydyn
- Nerosedyn
- Neufatin
- Neurodyn
- Neurosedin
- Neurosedym
- Neurosedyn
- Nevrodyn
- Nibrol
- Noctosediv
- Noxodyn
- Pangul
- Pantosediv
- Poly-Giron
- Polygripan
- Predni-Sediv
- Pro-ban M
- Profarmil
- Psycholiquid
- Psychotablets
- Quetimid
- Quietoplex
- Sandormin
- Sedalis
- Sedalis sedi-lab
- Sedimide
- Sedin
- Sedisperil
- Sedoval
- Shin-naito S
- Shinnibrol
- Sleepan
- Slipro
- Softenil
- Softenon
- THALOMID
- Talargan
- Talimol
- Talismol
- Telagan
- Telargan
- Telargean
- Tensival
- Thalidomide 99%
- Thalidomine USP26
- Thalin
- Thalinette
- Theophilcholine
- Ulcerfen
- Valgis
- Valgraine
- Yodomin
- alpha-phthalimidoglutarimide
Predni-Sediv Brand Names Mixture
- No information avaliable
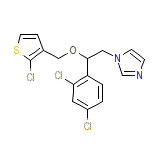
Predni-Sediv Chemical_Formula
C16H13Cl3N2OS
Predni-Sediv RX_link
http://www.rxlist.com/cgi/generic3/tioconaz.htm
Predni-Sediv fda sheet
Predni-Sediv msds (material safety sheet)
Predni-Sediv Synthesis Reference
No information avaliable
Predni-Sediv Molecular Weight
387.711 g/mol
Predni-Sediv Melting Point
No information avaliable
Predni-Sediv H2O Solubility
No information avaliable
Predni-Sediv State
Solid
Predni-Sediv LogP
4.971
Predni-Sediv Dosage Forms
Cream; Ointment; Suppository (each applicator-full provides approximately 4.6 grams of ointment containing 300 mg of tioconazole)
Predni-Sediv Indication
For the local treatment of vulvovaginal candidiasis (moniliasis).
Predni-Sediv Pharmacology
Tioconazole is a broad-spectrum imidazole antifungal agent that inhibits the growth of human pathogenic yeasts. Tioconazole exhibits fungicidal activity in vitro against Candida albicans, other species of the genus Candida, and against Torulopsis glabrata. Tioconazole prevents the growth and function of some fungal organisms by interfering with the production of substances needed to preserve the cell membrane. This drug is effective only for infections caused by fungal organisms. It will not work for bacterial or viral infections.
Predni-Sediv Absorption
Systemic absorption following a single intravaginal application of tioconazole in nonpregnant patients is negligible.
Predni-Sediv side effects and Toxicity
Symptoms of overdose include erythema, stinging, blistering, peeling, edema, pruritus, urticaria, burning,
Predni-Sediv Patient Information
Predni-Sediv Organisms Affected
Yeast and other fungi