Nabac




Nabac Brand names, Nabac Analogs
- AT-7
- Acigena
- Almederm
- Bilevon
- Bivelon
- Compound G-11
- Cotofilm
- Dermadex
- Dial
- Distodin
- E-Z Scrub
- Eleven
- Exofene
- Fascol
- Fesia-Sin
- Fomac
- Fostril
- G-Eleven
- G-II
- Gamophen
- Gamophene
- Germa-Medica
- HCP
- Hexa-Germ
- Hexabalm
- Hexachlorofen
- Hexachlorophane
- Hexachlorophen
- Hexachlorophene, Pharma
- Hexafen
- Hexascrub
- Hexazinone
- Hexide
- Hexophene
- Hexosan
- Isobac
- Isobac 20
- Nabac
- Nabac 25 Ec
- Neosept V
- Phiso-Scrub
- Phisodan
- Phisohex
- Pre-Op
- Rcra Waste Number U132
- Ritosept
- Scrubteam Surgical Spongebrush
- Septi-Soft
- Septisol
- Septofen
- Soy-Dome
- Ster-Zac
- Steral
- Steraskin
- Surgi-Cen
- Surgi-Cin
- Surofene
- Tersaseptic
- Trichlorophene
- Turgex
Nabac Brand Names Mixture
- Fosteen Dermatologic Shampoo (Hexachlorophene + Lanolin + Salicylic Acid + Sulfur)
- Hexaphenyl (Hexachlorophene + Isopropyl Alcohol)
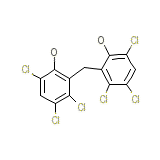
Nabac Chemical_Formula
C13H6Cl6O2
Nabac RX_link
http://www.rxlist.com/cgi/generic2/hexchlorph.htm
Nabac fda sheet
Nabac msds (material safety sheet)
Nabac Synthesis Reference
No information avaliable
Nabac Molecular Weight
406.902 g/mol
Nabac Melting Point
166.5 oC (boiling point)
Nabac H2O Solubility
140 mg/L
Nabac State
Liquid
Nabac LogP
6.941
Nabac Dosage Forms
Liquid
Nabac Indication
For use as a surgical scrub and a bacteriostatic skin cleanser. It may also be used to control an outbreak of gram-positive infection where other infection control procedures have been unsuccessful.
Nabac Pharmacology
Hexachlorophene, a detergent cleanser, is an antibacterial sudsing emulsion for topical administration. It is a bacteriostatic cleansing agent. It cleanses the skin thoroughly and has bacteriostatic action against staphylococci and other gram-positive bacteria. Cumulative antibacterial action develops with repeated use. Cleansing with alcohol or soaps containing alcohol removes the antibacterial residue.
Nabac Absorption
Detectable blood levels of hexachlorophene following absorption through intact skin have been found in subjects who regularly scrubbed with hexachlorophene.
Nabac side effects and Toxicity
Oral, rat LD50: 66 mg/kg. Signs of overdose include anorexia, vomiting, abdominal cramps, diarrhea, dehydration, convulsions, hypotension, and shock, and in several reported instances, fatalities.
Nabac Patient Information
No information avaliable
Nabac Organisms Affected
Enteric bacteria and other eubacteria