Sklero-tabuls




Sklero-tabuls Brand names, Sklero-tabuls Analogs
- Abitrate
- Amotril
- Amotril S
- Angiokapsul
- Anparton
- Antilipid
- Antilipide
- Apolan
- Arterioflexin
- Arterosol
- Artes
- Artevil
- Ateculon
- Ateriosan
- Athebrate
- Atheromide
- Atheropront
- Athranid-Wirkstoff
- Atrolen
- Atromid
- Atromid S
- Atromid-S
- Atromida
- Atromidin
- Atrovis
- Azionyl
- Bioscleran
- Bresit
- CPIB
- Cartagyl
- Chlorfenisate
- Chlorophenoxyisobutyricacidethylester
- Chlorphenisate
- Cinnarizin
- Cinnarizine
- Citiflus
- Claripex
- Claripex CPIB
- Cloberat
- Clobrat
- Clobren-5F
- Clobren-SF
- Clofar
- Clofibate
- Clofibram
- Clofibrat
- Clofibrato
- Clofibratum
- Clofinit
- Clofipront
- Delipid
- Deliva
- Dura clofibrat
- ELPI
- EPIB
- Ethyl chlorophenoxyisobutyrate
- Ethyl clofibrate
- Ethyl p-chlorophenoxyisobutyrate
- Ethyl para-chlorophenoxyisobutyrate
- Fibralem
- Gerastop
- Hyclorate
- Klofibrat
- Klofiran
- Levatrom
- Lipamid
- Lipavil
- Lipavlon
- Lipide 500
- Lipidsenker
- Lipofacton
- Lipomid
- Liponorm
- Liporeduct
- Liporil
- Liposid
- Liprin
- Liprinal
- Lobetrin
- Miscleron
- Negalip
- Neo-Atromid
- Normalip
- Normat
- Normet
- Normolipol
- Novofibrate
- Oxan 600
- Persantinat
- Recolip
- Regardin
- Regelan
- Regelan N
- Robigram
- Scrobin
- Serofinex
- Serotinex
- Skerolip
- Sklerepmexe
- Sklero
- Sklero-Tablinen
- Sklero-tablinene
- Sklero-tabuls
- Sklerolip
- Skleromex
- Skleromexe
- Tetrasipton
- Thillate
- Thiosan
- Ticlobran
- Vincamin compositum
- Xyduril
- Yoclo
- neo-Atomid
Sklero-tabuls Brand Names Mixture
- No information avaliable
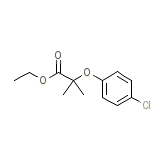
Sklero-tabuls Chemical_Formula
C12H15ClO3
Sklero-tabuls RX_link
http://www.rxlist.com/cgi/generic2/clofibrate.htm
Sklero-tabuls fda sheet
Sklero-tabuls msds (material safety sheet)
Sklero-tabuls Synthesis Reference
No information avaliable
Sklero-tabuls Molecular Weight
242.698 g/mol
Sklero-tabuls Melting Point
< 25 oC (boiling point 148-150°C at 25 mm Hg)
Sklero-tabuls H2O Solubility
Insoluble
Sklero-tabuls State
Liquid
Sklero-tabuls LogP
3.58
Sklero-tabuls Dosage Forms
Capsules (orange, oblong, soft-gelatin capsules, each containing 500 mg clofibrate)
Sklero-tabuls Indication
For Primary Dysbetalipoproteinemia (Type III hyperlipidemia) that does not respond adequately to diet.
Sklero-tabuls Pharmacology
Clofibrate is an antilipidemic agent similar to gemfibrozil. It acts to lower elevated serum lipids by reducing the very low-density lipoprotein fraction (Sf 20-400) rich in triglycerides. Serum cholesterol may be decreased, particularly in those patients whose cholesterol elevation is due to the presence of IDL as a result of Type III hyperlipoproteinemia. Several investigators have observed in their studies that clofibrate may produce a decrease in cholesterol linoleate but an increase in palmitoleate and oleate, the latter being considered atherogenic in experimental animals. The significance of this finding is unknown at this time. Reduction of triglycerides in some patients treated with clofibrate or certain of its chemically and clinically similar analogs may be associated with an increase in LDL cholesterol. Increase in LDL cholesterol has been observed in patients whose cholesterol is initially normal. Animal studies suggest that clofibrate interrupts cholesterol biosynthesis prior to mevalonate formation.
Sklero-tabuls Absorption
Completely but slowly absorbed from the intestine. Between 95% and 99% of an oral dose of clofibrate is excreted in the urine as free and conjugated clofibric acid; thus, the absorption of clofibrate is virtually complete.
Sklero-tabuls side effects and Toxicity
Oral, mouse: LD50 = 1220 mg/kg; Oral, rabbit: LD50 = 1370 mg/kg; Oral, rat: LD50 = 940 mg/kg. No reported case of overdosage in humans.
Sklero-tabuls Patient Information
Sklero-tabuls Organisms Affected
Humans and other mammals