Pedi-Dri




Pedi-Dri Brand names, Pedi-Dri Analogs
Pedi-Dri Brand Names Mixture
- No information avaliable
Pedi-Dri Chemical_Formula
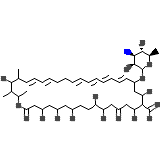
C47H75NO17
Pedi-Dri RX_link
http://www.rxlist.com/cgi/generic2/nystat.htm
Pedi-Dri fda sheet
Pedi-Dri msds (material safety sheet)
Pedi-Dri Synthesis Reference
No information avaliable
Pedi-Dri Molecular Weight
926.09 g/mol
Pedi-Dri Melting Point
160oC
Pedi-Dri H2O Solubility
360 mg/L
Pedi-Dri State
Solid
Pedi-Dri LogP
-0.534
Pedi-Dri Dosage Forms
Cream; Drops; Liquid; Ointment; Suspension; Tablet
Pedi-Dri Indication
For treatment of cutaneous or mucocutaneous mycotic infections caused by Candida species
Pedi-Dri Pharmacology
Nystatin is an antibiotic which is both fungistatic and fungicidal in vitro against a wide variety of yeasts and yeast-like fungi, including Candida albicans, C. parapsilosis, C. tropicalis, C. guilliermondi, C. pseudotropicalis, C. krusei, Torulopsis glabrata, Tricophyton rubrum, T. mentagrophytes. Nystatin acts by binding to sterols in the cell membrane of susceptible species resulting in a change in membrane permeability and the subsequent leakage of intracellular components. On repeated subculturing with increasing levels of nystatin, Candida albicans does not develop resistance to nystatin. Generally, resistance to nystatin does not develop during therapy. However, other species of Candida (C. tropicalis, C. guilliermondi, C. krusei, and C. stellatoides) become quite resistant on treatment with nystatin and simultaneously become cross resistant to amphotericin as well. This resistance is lost when the antibiotic is removed. Nystatin exhibits no appreciable activity against bacteria, protozoa, or viruses.
Pedi-Dri Absorption
Nystatin is not absorbed from intact skin or mucous membrane
Pedi-Dri side effects and Toxicity
No information avaliable
Pedi-Dri Patient Information
No information avaliable
Pedi-Dri Organisms Affected
Fungi