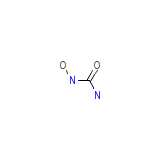
Hydroxicarbamidum




Hydroxicarbamidum Brand names, Hydroxicarbamidum Analogs
- Biosupressin
- Carbamohydroxamic Acid
- Carbamohydroximic Acid
- Carbamohydroxyamic Acid
- Carbamoyl Oxime
- Carbamyl Hydroxamate
- Droxia
- HU
- Hidrix
- Hydrea
- Hydreia
- Hydroxicarbamidum
- Hydroxycarbamide
- Hydroxycarbamine
- Hydroxylurea
- Hydura
- Hydurea
- Idrossicarbamide [Dcit]
- Litaler
- Litalir
- N-Carbamoylhydroxylamine
- N-Hydroxyurea
- Onco-Carbide
- Oxyurea
- Sterile Urea
- Ureaphil
Hydroxicarbamidum Brand Names Mixture
- No information avaliable
Hydroxicarbamidum Chemical_Formula
CH4N2O2
Hydroxicarbamidum RX_link
http://www.rxlist.com/cgi/generic2/hydroxyurea.htm
Hydroxicarbamidum fda sheet
Hydroxicarbamidum msds (material safety sheet)
Hydroxicarbamidum Synthesis Reference
No information avaliable
Hydroxicarbamidum Molecular Weight
76.0547 g/mol
Hydroxicarbamidum Melting Point
142-146 oC
Hydroxicarbamidum H2O Solubility
1E+006 mg/L
Hydroxicarbamidum State
Solid
Hydroxicarbamidum LogP
-1.432
Hydroxicarbamidum Dosage Forms
Capsule
Hydroxicarbamidum Indication
For management of melanoma, resistant chronic myelocytic leukemia, and recurrent, metastatic, or inoperable carcinoma of the ovary and Sickle-cell anemia.
Hydroxicarbamidum Pharmacology
Hydroxyurea has dose-dependent synergistic activity with cisplatin in vitro. In vivo Hydroxyurea showed activity in combination with cisplatin against the LX-1 and CALU-6 human lung xenografts, but minimal activity was seen with the NCI-H460 or NCI-H520 xenografts. Hydroxyurea was synergistic with cisplatin in the Lewis lung murine xenograft. Sequential exposure to Hydroxyurea 4 hours before cisplatin produced the greatest interaction.
Hydroxicarbamidum Absorption
Well absorbed from the gastrointestinal tract.
Hydroxicarbamidum side effects and Toxicity
Oral, mouse: LD50 = 7330 mg/kg; Oral, rat: LD50 = 5760 mg/kg
Hydroxicarbamidum Patient Information
HYDREA is a medication that must be handled with care. People who are not taking HYDREA should not be exposed to it. If the powder from the capsule is spilled, it should be wiped up immediately with a damp disposable towel and discarded in a closed container, such as a plastic bag. The medication should be kept away from children and pets.
Hydroxicarbamidum Organisms Affected
Humans and other mammals