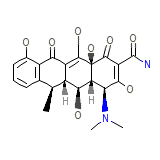
Doxycycline Hyclate




Doxycycline Hyclate Brand names, Doxycycline Hyclate Analogs
Doxycycline Hyclate Brand Names Mixture
- No information avaliable
Doxycycline Hyclate Chemical_Formula
C22H24N2O8
Doxycycline Hyclate RX_link
http://www.rxlist.com/cgi/generic/doxycyc.htm
Doxycycline Hyclate fda sheet
Doxycycline Hyclate msds (material safety sheet)
Doxycycline Hyclate Synthesis Reference
C. R. Stephens et al.; J. Am. Chem. Soc. 80; 5324 (1958)
Doxycycline Hyclate Molecular Weight
444.435 g/mol
Doxycycline Hyclate Melting Point
201 oC
Doxycycline Hyclate H2O Solubility
630 mg/L
Doxycycline Hyclate State
Solid
Doxycycline Hyclate LogP
-1.219
Doxycycline Hyclate Dosage Forms
Tablet; Capsule; Gel; Liquid; Powder; Powder for solution
Doxycycline Hyclate Indication
For the treatment of the following infections:Rocky mountain spotted fever, typhus fever and the typhus group, Q fever, rickettsialpox, and tick fevers caused by Rickettsiae, Chancroid caused by Haemophilus ducreyi, Uncomplicated gonorrhea caused by Neisseria gonorrhoeae, Respiratory tract infections caused by Mycoplasma pneumoniae or Streptococcus pneumoniae. Lymphogranuloma ve
Doxycycline Hyclate Pharmacology
Doxycycline, a long-acting tetracycline derived from oxytetracycline, is used to inhibit bacterial protein synthesis and treat non-gonococcal urethritis and cervicitis, exacerbations of bronchitis in patients with COPD, and adult periodontitis.
Doxycycline Hyclate Absorption
completely absorbed after oral administration
Doxycycline Hyclate side effects and Toxicity
anorexia, nausea, diarrhoea, glossitis, dysphagia, enterocolitis and inflammatory lesions (with monilial overgrowth) in the anogenital region. skin reactions such as maculopapular and erythematous rashes, exfoliative dermatitis, photosensitivity, hypersensitivity reactions such as urticaria, angioneurotic oedema, anaphylaxis, anaphyl-actoid purpura, pericarditis, and exacerbation of systemic lupus erythematosus, benign intracranial hypertension in adults disappearing on discontinuation of the medicine, haematologic abnormalities such as haemolytic anaemia, thrombocytopenia, neutropenia, and eosinophilia. LD50=262 mg/kg(i.p. in rat)
Doxycycline Hyclate Patient Information
Doxycycline Hyclate Organisms Affected
Enteric bacteria and other eubacteria