ASTA




ASTA Brand names, ASTA Analogs
ASTA Brand Names Mixture
- No information avaliable
ASTA Chemical_Formula
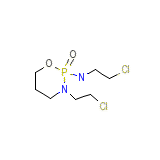
C7H15Cl2N2O2P
ASTA RX_link
http://www.rxlist.com/cgi/generic2/ifosfamide.htm
ASTA fda sheet
ASTA msds (material safety sheet)
ASTA Synthesis Reference
No information avaliable
ASTA Molecular Weight
261.085 g/mol
ASTA Melting Point
39-41 oC
ASTA H2O Solubility
3780 mg/L
ASTA State
Solid
ASTA LogP
-0.01
ASTA Dosage Forms
Powder for constitution and administration by intravenous infusion
ASTA Indication
For third line chemotherapy of germ cell testicular cancer. It should ordinarily be used in combination with a prophylactic agent for hemorrhagic cystitis, such as mesna.
ASTA Pharmacology
Ifosfamide selectively inhibits the synthesis of deoxyribonucleic acid (DNA). The guanine and cytosine content correlates with the degree of ifosfamide-induced cross-linking. At high concentrations of the drug, cellular RNA and protein synthesis are also suppressed.
ASTA Absorption
No information avaliable
ASTA side effects and Toxicity
LD50 (mouse) = 390-1005 mg/kg, LD50 (rat) = 150-190 mg/kg. Side effects include nausea, vomiting and myelosuppression. Toxic effects include central nervous system toxicity (confusion, hallucinations) and urotoxic effects (cystitis, blood in urine).
ASTA Patient Information
No information avaliable
ASTA Organisms Affected
Humans and other mammals