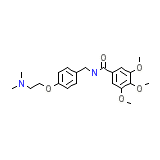
Anvitoff




Anvitoff Brand names, Anvitoff Analogs
Anvitoff Brand Names Mixture
- No information avaliable
Anvitoff Chemical_Formula
C21H28N2O5
Anvitoff RX_link
http://www.rxlist.com/cgi/generic3/trimethbenz.htm
Anvitoff fda sheet
Anvitoff msds (material safety sheet)
Anvitoff Synthesis Reference
No information avaliable
Anvitoff Molecular Weight
388.458 g/mol
Anvitoff Melting Point
188.7 oC
Anvitoff H2O Solubility
40 mg/L
Anvitoff State
Solid
Anvitoff LogP
2.784
Anvitoff Dosage Forms
Capsules (300 mg) for oral administration; Suppositories (200 mg); Injectable (100 mg, 200 mg)
Anvitoff Indication
For the treatment of postoperative nausea and vomiting and for nausea associated with gastroenteritis.
Anvitoff Pharmacology
Trimethobenzamide is a novel antiemetic which prevents nausea and vomiting in humans. Its actions are unclear but most likely involves the chemoreceptor trigger zone (CTZ). In dogs pretreated with trimethobenzamide HCl, the emetic response to apomorphine is inhibited, while little or no protection is afforded against emesis induced by intragastric copper sulfate.
Anvitoff Absorption
The relative bioavailability of the capsule formulation compared to the solution is 100%.
Anvitoff side effects and Toxicity
Oral LD50 in mice is 1600 mg/kg.
Anvitoff Patient Information
No information avaliable
Anvitoff Organisms Affected
Humans and other mammals