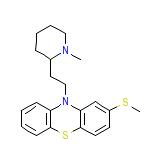
Stalleril




Stalleril Brand names, Stalleril Analogs
- Aldazine
- Mallorol
- Malloryl
- Meleril
- Mellaril
- Mellaril Hydrochloride
- Mellaril-S
- Mellarit
- Mellerets
- Mellerette
- Melleretten
- Melleril
- Metlaril
- Novoridazine
- Orsanil
- Ridazin
- Ridazine
- Sonapax
- Sonapax Hydrochloride
- Stalleril
- Thioridazin
- Thioridazine Chloride
- Thioridazine Hcl
- Thioridazine Hcl Intensol
- Thioridazine Hydrochloride
- Thioridazine Hydrochloride [Jan]
- Thioridazine, Prolongatum
- Thioridazinhydrochlorid
- Thoridazine Hydrochloride
- Tioridazin
- Usaf Sz-3
- Usaf Sz-B
Stalleril Brand Names Mixture
- No information avaliable
Stalleril Chemical_Formula
C21H26N2S2
Stalleril RX_link
http://www.rxlist.com/cgi/generic3/thioridazine.htm
Stalleril fda sheet
Stalleril msds (material safety sheet)
Stalleril Synthesis Reference
Bourquin et al., Helv. Chim. Acta 41, 1072 (1958)
Stalleril Molecular Weight
370.577 g/mol
Stalleril Melting Point
73 oC
Stalleril H2O Solubility
0.0336 mg/L
Stalleril State
Solid
Stalleril LogP
6.552
Stalleril Dosage Forms
Suspension; Tablet
Stalleril Indication
For the treatment of schizophrenia and generalized anxiety disorder.
Stalleril Pharmacology
Thioridazine is a trifluoro-methyl phenothiazine derivative intended for the management of schizophrenia and other psychotic disorders. Thioridazine has not been shown effective in the management of behaviorial complications in patients with mental retardation.
Stalleril Absorption
60%
Stalleril side effects and Toxicity
LD50=956-1034 mg/kg (Orally in rats); Agitation, blurred vision, coma, confusion, constipation, difficulty breathing, dilated or constricted pupils, diminished flow of urine, dry mouth, dry skin, excessively high or low body temperature, extremely low blood pressure, fluid in the lungs, heart abnormalities, inability to urinate, intestinal blockage, nasal congestion, restlessness, sedation, seizures, shock
Stalleril Patient Information
Stalleril Organisms Affected
Humans and other mammals