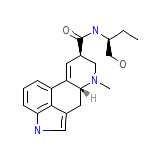
Ryegonovin




Ryegonovin Brand names, Ryegonovin Analogs
Ryegonovin Brand Names Mixture
- No information avaliable
Ryegonovin Chemical_Formula
C20H25N3O2
Ryegonovin RX_link
http://www.rxlist.com/cgi/generic2/methylerg.htm
Ryegonovin fda sheet
Ryegonovin msds (material safety sheet)
Ryegonovin Synthesis Reference
A. Stoll et al., eidem, Helv. Chim. Acta. 26, 944 (1943)
Ryegonovin Molecular Weight
339.432 g/mol
Ryegonovin Melting Point
172 oC
Ryegonovin H2O Solubility
25 mg/mL
Ryegonovin State
Solid
Ryegonovin LogP
1.01
Ryegonovin Dosage Forms
Injection (intramuscular or intravenous); Tablet
Ryegonovin Indication
For the prevention and control of excessive bleeding following vaginal childbirth
Ryegonovin Pharmacology
Methylergonovine is a semisynthetic ergot alkaloid and a derivative of ergonovine and is used for the prevention and control of postpartum and post-abortion hemorrhage. In general, the effects of all the ergot alkaloids appear to results from their actions as partial agonists or antagonists at adrenergic, dopaminergic, and tryptaminergic receptors. The spectrum of effects depends on the agent, dosage, species, tissue, and experimental or physiological conditions. All of the alkaloids of ergot significantly increase the motor activity of the uterus. After small doses contractions are increased in force or frequency, or both, but are followed by a normal degree of relaxation. As the dose is increased, contractions become more forceful and prolonged, resting tonus is markedly increased, and sustained contracture can result.
Ryegonovin Absorption
Absorption is rapid after oral (60% bioavailability) and intramuscular (78% bioavailability) administration.
Ryegonovin side effects and Toxicity
Signs and symptoms of overexposure: hypertension, seizures, headache, hypotension, nausea, and vomiting.
Ryegonovin Patient Information
Ryegonovin Organisms Affected
Humans and other mammals