Liskantin




Liskantin Brand names, Liskantin Analogs
- Apo-Primidone
- Cyral
- Desoxyphenobarbitone
- Hexadiona
- Hexamidine
- Lepimidin
- Lepsiral
- Liskantin
- Majsolin
- Medi-Pets
- Midone
- Milepsin
- Misodine
- Misolyne
- Mizodin
- Mizolin
- Myidone
- Mylepsin
- Mylepsinum
- Mysedon
- Mysoline
- Neurosyn
- Pms Primidone
- Prilepsin
- Primacione
- Primaclone
- Primacone
- Primakton
- Primidon
- Primidone Methanol Solution
- Primoline
- Prysoline
- Pyrimidone "Medi-Pets"
- Pyrimidone Medi-Pets
- Resimatil
- Sertan
Liskantin Brand Names Mixture
- No information avaliable
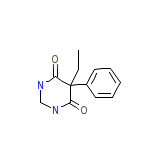
Liskantin Chemical_Formula
C12H14N2O2
Liskantin RX_link
http://www.rxlist.com/cgi/generic2/primid.htm
Liskantin fda sheet
Liskantin msds (material safety sheet)
Liskantin Synthesis Reference
Boon et al., Brit. pat. 666,027 (1952 to I.C.I.)
Liskantin Molecular Weight
218.252 g/mol
Liskantin Melting Point
281.5 oC
Liskantin H2O Solubility
500 mg/L
Liskantin State
Solid
Liskantin LogP
1.713
Liskantin Dosage Forms
Tablet; Chewable tablets; Oral suspension
Liskantin Indication
For the treatment of epilepsy
Liskantin Pharmacology
Primidone is a barbiturate with anticonvulsant properties. Primidone, either alone or used concomitantly with other anticonvulsants, is indicated in the control of grand mal, psychomotor, and focal epileptic seizures. It may control grand mal seizures refractory to other anticonvulsant therapy. Primidone raises electro- or chemoshock seizure thresholds or alters seizure patterns in experimental animals. Primidone per se has anticonvulsant activity as do its two metabolites, phenobarbital and phenylethylmalonamide (PEMA). In addition to its anticonvulsant activity, Primidone potentiates that of phenobarbital in experimental animals.
Liskantin Absorption
90 to 100%
Liskantin side effects and Toxicity
No information avaliable
Liskantin Patient Information
Liskantin Organisms Affected
Humans and other mammals