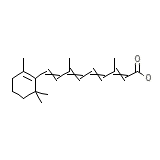
Tri-Luma




Tri-Luma Brand names, Tri-Luma Analogs
- Aberel
- Accutane
- Airol
- Aknefug
- Aknoten
- All Trans Retinoic Acid
- All Trans-Retinoic Acid
- Amnesteem
- Avita
- Beta-Retinoic Acid
- Claravis
- Dermairol
- Eudyna
- Isotretinoin
- Lsotretinoin
- Panretin
- Renova
- Retin-A
- Retin-a Micro
- Retinoate
- Retinoic Acid
- Retinoic Acid, All Trans Isomer
- Retionic Acid
- Retisol-A
- Solage
- Sotret
- Stieva-A
- Stieva-a Forte
- Trans-Retinoic Acid
- Tretin M
- Tri-Luma
- Vesanoid
- Vitamin a Acid
- Vitinoin
- ??-Retinoic Acid
Tri-Luma Brand Names Mixture
- No information avaliable
Tri-Luma Chemical_Formula
C20H28O2
Tri-Luma RX_link
http://www.rxlist.com/cgi/generic/tretinoin.htm
Tri-Luma fda sheet
Tri-Luma msds (material safety sheet)
Tri-Luma Synthesis Reference
Matsui et al.; J.Vitaminol.; 4; 190, 192(1958)
Tri-Luma Molecular Weight
300.435 g/mol
Tri-Luma Melting Point
181 oC
Tri-Luma H2O Solubility
<0.1 g/100 mL
Tri-Luma State
Solid
Tri-Luma LogP
5.067
Tri-Luma Dosage Forms
Capsule; Cream; Gel; Liquid
Tri-Luma Indication
For the the induction of remission in patients with acute promyelocytic leukemia (APL), French-American-British (FAB) classification M3 (including the M3 variant); For the topical treatment of acne vulgaris, flat warts and other skin conditions (psoriasis, ichthyosis congenita, icthyosis vulgaris, lamellar icthyosis, keratosis palmaris et plantaris, epidermolytic hyperkeratosis, senile comedones, senile keratosis, keratosis follicularis (Darier's disease), and basal cell carcinomas.); For palliative therapy to improve fine wrinkling, mottled hyperpigmentation, roughness associated with photodamage.
Tri-Luma Pharmacology
Tretinoin, also known as all-trans-retinoic acid (ATRA), is a naturally occurring derivative of vitamin A (retinol). Retinoids such as tretinoin are important regulators of cell reproduction, proliferation, and differentiation and are used to treat acne and photodamaged skin and to manage keratinization disorders such as ichthyosis and keratosis follicularis. Tretinoin also represents the class of anticancer drugs called differentiating agents and is used in the treatment of acute promyelocytic leukemia (APL).
Tri-Luma Absorption
1-31% (topical);
Tri-Luma side effects and Toxicity
No information avaliable
Tri-Luma Patient Information
No information avaliable
Tri-Luma Organisms Affected
Humans and other mammals