Zovirax Wellstat Pac




Zovirax Wellstat Pac Brand names, Zovirax Wellstat Pac Analogs
Zovirax Wellstat Pac Brand Names Mixture
- Zovirax Injection (Zovirax sodium)
Zovirax Wellstat Pac Chemical_Formula
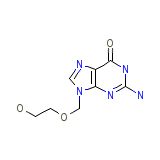
C8H11N5O3
Zovirax Wellstat Pac RX_link
http://www.rxlist.com/cgi/generic/acyclo.htm
Zovirax Wellstat Pac fda sheet
Zovirax Wellstat Pac msds (material safety sheet)
Zovirax Wellstat Pac Synthesis Reference
H. Matsumoto et al., Chem. Pharm. Bull. 35, 1153 (1988)
Zovirax Wellstat Pac Molecular Weight
225.205 g/mol
Zovirax Wellstat Pac Melting Point
256.5 - 257 oC
Zovirax Wellstat Pac H2O Solubility
3.399 mg/L
Zovirax Wellstat Pac State
Solid
Zovirax Wellstat Pac LogP
-1.314
Zovirax Wellstat Pac Dosage Forms
Intravenous infusion; Capsule; Tablet; Suspension; Topical cream; Topical ointment
Zovirax Wellstat Pac Indication
For the treatment and management of herpes zoster (shingles), genital herpes, and chickenpox
Zovirax Wellstat Pac Pharmacology
Acyclovir is a synthetic deoxyguanosine analog and it is the prototype antiviral agent that is activated by viral thymidine kinase. The selective activity of acyclovir is due to its affinity for the thymidine kinase enzyme encoded by HSV and VZV.
Zovirax Wellstat Pac Absorption
Oral: bioavailability 10 to 20%
Zovirax Wellstat Pac side effects and Toxicity
Acyclovir may cause nephrotoxicity (crystallization of acyclovir within renal tubules, elevation of serum creatinine, transient), and neurotoxicity (coma, hallucinations, lethargy, seizures, tremors). Nephrotoxicity and neurotoxicity usually resolve after cessation of acyclovir therapy. However, there is no well-defined relationship between acyclovir concentrations in the blood and these adverse effects.
Zovirax Wellstat Pac Patient Information
Zovirax Wellstat Pac Organisms Affected
Human herpes virus