Oradil




Oradil Brand names, Oradil Analogs
Oradil Brand Names Mixture
- Tenoretic (chlorthalidone + atenolol)
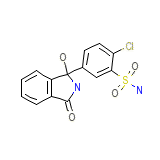
Oradil Chemical_Formula
C14H11ClN2O4S
Oradil RX_link
http://www.rxlist.com/cgi/generic3/chlorthalidone.htm
Oradil fda sheet
Oradil msds (material safety sheet)
Oradil Synthesis Reference
Graf et al.; Helv. Chem.; Acta 42; 1085 (1959); U.S.Pat.3,055,904 (1962)
Oradil Molecular Weight
338.767 g/mol
Oradil Melting Point
218-264oC
Oradil H2O Solubility
120 mg/L
Oradil State
Solid
Oradil LogP
1.718
Oradil Dosage Forms
Tablet
Oradil Indication
For management of hypertension either as the sole therapeutic agent or to enhance the effect of other antihypertensive drugs in the more severe forms of hypertension.
Oradil Pharmacology
Chlorthalidone, a monosulfonamyl diuretic, differs form other thiazide diuretics in that a double ring system is incorporated into its structure. Chlorthalidone is used alone or with atenolol in the management of hypertension and edema.
Oradil Absorption
Absorbed relatively rapidly after oral administration.
Oradil side effects and Toxicity
Symptoms of overdose include nausea, weakness, dizziness and disturbances of electrolyte balance.
Oradil Patient Information
Patients should inform their physician if they have: (1) had an allergic reaction to chlorthalidone or other diuretics or have asthma, (2) kidney disease, (3) liver disease, (4) gout, (5) systemic lupus erythematosus, or (6) been taking other drugs such as cortisone, digitalis, lithium carbonate, or drugs for diabetes.
Patients should be cautioned to contact their physician if they experience any of the following symptoms of potassium loss: excess thirst, tiredness, drowsiness, restlessness, muscle pains or cramps, nausea, vomiting, or increased heart rate or pulse.
Patients should also be cautioned that taking alcohol can increase the chance of dizziness occurring.
Oradil Organisms Affected
Humans and other mammals