Megestat




Megestat Brand names, Megestat Analogs
Megestat Brand Names Mixture
- No information avaliable
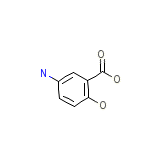
Megestat Chemical_Formula
C7H7NO3
Megestat RX_link
http://www.rxlist.com/cgi/generic2/canasa.htm
Megestat fda sheet
Megestat msds (material safety sheet)
Megestat Synthesis Reference
No information avaliable
Megestat Molecular Weight
153.135 g/mol
Megestat Melting Point
283 oC
Megestat H2O Solubility
0.84 g/L at 20°C
Megestat State
Solid
Megestat LogP
1.012
Megestat Dosage Forms
Rectal suppository (500 mg or 1000 mg)
Megestat Indication
For the treatment of active ulcerative proctitis.
Megestat Pharmacology
Sulfasalazine has been used in the treatment of ulcerative colitis for over 55 years. It is split by bacterial action in the colon into sulfapyridine (SP) and mesalamine (5-ASA). It is thought that the mesalamine component only is therapeutically active in ulcerative colitis.
Megestat Absorption
20 to 30% absorbed following oral administration. 10 to 35% absorbed from the colon (rectal suppository) - extent of absorption is determined by the length of time the drug is retained in the colon.
Megestat side effects and Toxicity
Oral, mouse: LD50 = 3370 mg/kg; Oral, rat: LD50 = 2800 mg/kg; Skin, rabbit: LD50 = >5 gm/kg. There have been no documented reports of serious toxicity in man resulting from massive overdosing with mesalamine. Under ordinary circumstances, mesalamine absorption from the colon is limited.
Megestat Patient Information
Megestat Organisms Affected
Humans and other mammals