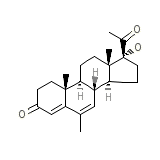
meso-NDGA




meso-NDGA Brand names, meso-NDGA Analogs
meso-NDGA Brand Names Mixture
- No information avaliable
meso-NDGA Chemical_Formula
C22H30O3
meso-NDGA RX_link
http://www.rxlist.com/cgi/generic4/megace_es.htm
meso-NDGA fda sheet
meso-NDGA msds (material safety sheet)
meso-NDGA Synthesis Reference
No information avaliable
meso-NDGA Molecular Weight
342.472 g/mol
meso-NDGA Melting Point
218.0-220.0oC
meso-NDGA H2O Solubility
2 µg/mL (at 37°C for the acetate salt)
meso-NDGA State
Solid
meso-NDGA LogP
3.123
meso-NDGA Dosage Forms
Oral suspension containing 125 mg of megestrol acetate per mL; Tablet
meso-NDGA Indication
For the treatment of anorexia, cachexia, or an unexplained, significant weight loss in patients with a diagnosis of acquired immunodeficiency syndrome (AIDS). Also used to treat breast cancer, endometrial cancer, and prostate cancer in Canada and some other countries.
meso-NDGA Pharmacology
Megestrol is a synthetic progestin and has the same physiologic effects as natural progesterone. The precise mechanism of megestrol’s antianorexic and anticachetic effects is unknown. Initially developed as a contraceptive, it was first evaluated in breast cancer treatment in 1967.
meso-NDGA Absorption
Variable, but well absorbed orally.
meso-NDGA side effects and Toxicity
No serious unexpected side effects have resulted from studies involving megestrol acetate oral suspension administered in dosages as high as 1200 mg/day.
meso-NDGA Patient Information
No information avaliable
meso-NDGA Organisms Affected
Humans and other mammals