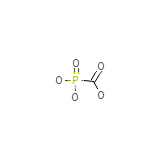
Phosphonoformate




Phosphonoformate Brand names, Phosphonoformate Analogs
Phosphonoformate Brand Names Mixture
- No information avaliable
Phosphonoformate Chemical_Formula
CH3O5P
Phosphonoformate RX_link
http://www.rxlist.com/cgi/generic2/foscarnet.htm
Phosphonoformate fda sheet
Phosphonoformate msds (material safety sheet)
Phosphonoformate Synthesis Reference
Noren, Jan O.; et al., J.Med.Chem. 26(2) 264-270(1983)
Phosphonoformate Molecular Weight
126.005 g/mol
Phosphonoformate Melting Point
88.06 oC
Phosphonoformate H2O Solubility
Complete
Phosphonoformate State
Solid
Phosphonoformate LogP
No information avaliable
Phosphonoformate Dosage Forms
Injection
Phosphonoformate Indication
For the treatment of CMV retinitis in patients with acquired immunodeficiency syndrome (AIDS) and for treatment of acyclovir-resistant mucocutaneous HSV infections in immunocompromised patients.
Phosphonoformate Pharmacology
Foscarnet is an organic analogue of inorganic pyrophosphate that inhibits replication of herpes viruses in vitro including cytomegalovirus (CMV) and herpes simplex virus types 1 and 2 (HSV-1 and HSV-2). Foscarnet does not require activation (phosphorylation) by thymidine kinase or other kinases and therefore is active in vitro against HSV TK deficient mutants and CMV UL97 mutants. Thus, HSV strains resistant to acyclovir or CMV strains resistant to ganciclovir may be sensitive to foscarnet. However, acyclovir or ganciclovir resistant mutants with alterations in the viral DNA polymerase may be resistant to foscarnet and may not respond to therapy with foscarnet. The combination of foscarnet and ganciclovir has been shown to have enhanced activity in vitro.
Phosphonoformate Absorption
Poorly absorbed after oral administration (bioavailability from 12 to 22%).
Phosphonoformate side effects and Toxicity
Oral, rat LD50: >2,000 mg/kg. Signs of overdose include renal impairment.
Phosphonoformate Patient Information
Phosphonoformate Organisms Affected
Human herpes viruses