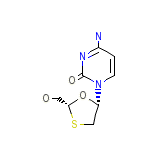
Epivir-Hbv




Epivir-Hbv Brand names, Epivir-Hbv Analogs
Epivir-Hbv Brand Names Mixture
- Combivir (Lamivudine + Zidovudine)
- Kivexa (Abacavir (Abacavir Sulfate) + Lamivudine)
- Trizivir (Abacavir (Abacavir Sulfate) + Lamivudine + Zidovudine)
Epivir-Hbv Chemical_Formula
C8H11N3O3S
Epivir-Hbv RX_link
http://www.rxlist.com/cgi/generic3/lamivudine.htm
Epivir-Hbv fda sheet
Epivir-Hbv msds (material safety sheet)
Epivir-Hbv Synthesis Reference
No information avaliable
Epivir-Hbv Molecular Weight
229.257 g/mol
Epivir-Hbv Melting Point
160-162 oC
Epivir-Hbv H2O Solubility
70 mg/ml
Epivir-Hbv State
Solid
Epivir-Hbv LogP
No information avaliable
Epivir-Hbv Dosage Forms
Solution; Tablet
Epivir-Hbv Indication
For the treatment of HIV infection and chronic hepatitis B (HBV).
Epivir-Hbv Pharmacology
Lamivudine is a nucleoside reverse transcriptase inhibitor (NRTI) with activity against Human Immunodeficiency Virus Type 1 (HIV-1) and hepatitis B (HBV). Lamivudine is phosphorylated to active metabolites that compete for incorporation into viral DNA. They inhibit the HIV reverse transcriptase enzyme competitively and act as a chain terminator of DNA synthesis. The lack of a 3'-OH group in the incorporated nucleoside analogue prevents the formation of the 5' to 3' phosphodiester linkage essential for DNA chain elongation, and therefore, the viral DNA growth is terminated.
Epivir-Hbv Absorption
Lamivudine was rapidly absorbed after oral administration in HIV-infected patients. Absolute bioavailability in adults is 86% ± 16% for the tablet and 87% ± 13% for the oral solution.
Epivir-Hbv side effects and Toxicity
No information avaliable
Epivir-Hbv Patient Information
Epivir-Hbv Organisms Affected
Human immunodeficiency virus and Hepatitis B virus