Trizivir




Trizivir Brand names, Trizivir Analogs
Trizivir Brand Names Mixture
- Combivir (Lamivudine + Zidovudine)
- Kivexa (Abacavir (Abacavir Sulfate) + Lamivudine)
- Trizivir (Abacavir (Abacavir Sulfate) + Lamivudine + Zidovudine)
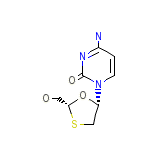
Trizivir Chemical_Formula
C8H11N3O3S
Trizivir RX_link
http://www.rxlist.com/cgi/generic3/lamivudine.htm
Trizivir fda sheet
Trizivir msds (material safety sheet)
Trizivir Synthesis Reference
No information avaliable
Trizivir Molecular Weight
229.257 g/mol
Trizivir Melting Point
160-162 oC
Trizivir H2O Solubility
70 mg/ml
Trizivir State
Solid
Trizivir LogP
No information avaliable
Trizivir Dosage Forms
Solution; Tablet
Trizivir Indication
For the treatment of HIV infection and chronic hepatitis B (HBV).
Trizivir Pharmacology
Lamivudine is a nucleoside reverse transcriptase inhibitor (NRTI) with activity against Human Immunodeficiency Virus Type 1 (HIV-1) and hepatitis B (HBV). Lamivudine is phosphorylated to active metabolites that compete for incorporation into viral DNA. They inhibit the HIV reverse transcriptase enzyme competitively and act as a chain terminator of DNA synthesis. The lack of a 3'-OH group in the incorporated nucleoside analogue prevents the formation of the 5' to 3' phosphodiester linkage essential for DNA chain elongation, and therefore, the viral DNA growth is terminated.
Trizivir Absorption
Lamivudine was rapidly absorbed after oral administration in HIV-infected patients. Absolute bioavailability in adults is 86% ± 16% for the tablet and 87% ± 13% for the oral solution.
Trizivir side effects and Toxicity
No information avaliable
Trizivir Patient Information
Trizivir Organisms Affected
Human immunodeficiency virus and Hepatitis B virus