Rifadin Iv




Rifadin Iv Brand names, Rifadin Iv Analogs
Rifadin Iv Brand Names Mixture
- Rifater (Isoniazid + Pyrazinaamide + Rifampin)
- Rifamate (Rifampin + Isoniazid)
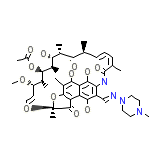
Rifadin Iv Chemical_Formula
Rifadin Iv RX_link
Rifadin Iv fda sheet
Rifadin Iv msds (material safety sheet)
Rifadin Iv Synthesis Reference
Rifadin Iv Molecular Weight
Rifadin Iv Melting Point
Rifadin Iv H2O Solubility
Rifadin Iv State
Rifadin Iv LogP
Rifadin Iv Dosage Forms
Rifadin Iv Indication
Rifadin Iv Pharmacology
Rifadin Iv Absorption
Rifadin Iv side effects and Toxicity
Rifadin Iv Patient Information
The patient should be told that rifampin may produce a reddish coloration of the urine, sweat, sputum, and tears, and the patient should be forewarned of this. Soft contact lenses may be permanently stained. The patients should be advised that the reliability of oral or other systemic hormonal contraceptives may be affected; consideration should be given to using alternative contraceptive measures. Patients should be instructed to take rifampin either 1 hour before or 2 hours after a meal with a full glass of water. Patients should be instructed to notify their physicians promptly if they experience any of the following: fever, loss of appetite, malaise, nausea and vomiting, darkened urine, yellowish discoloration of the skin and eyes, and pain or swelling of the joints. Compliance with the full course of therapy must be emphasized, and the importance of not missing any doses must be stressed.