Imidourea




Imidourea Brand names, Imidourea Analogs
Imidourea Brand Names Mixture
- No information avaliable
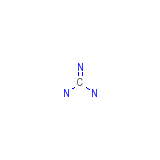
Imidourea Chemical_Formula
CH5N3
Imidourea RX_link
No information avaliable
Imidourea fda sheet
Imidourea msds (material safety sheet)
Imidourea Synthesis Reference
No information avaliable
Imidourea Molecular Weight
59.0706 g/mol
Imidourea Melting Point
183 oC (HCl salt)
Imidourea H2O Solubility
500 mg/mL (HCl salt)
Imidourea State
Solid
Imidourea LogP
-0.946
Imidourea Dosage Forms
No information avaliable
Imidourea Indication
For the reduction of the symptoms of muscle weakness and easy fatigability associated with the myasthenic syndrome of Eaton-Lambert. It is not indicated for treating myasthenia gravis.
Imidourea Pharmacology
Guanidine apparently acts by enhancing the release of acetylcholine following a nerve impulse. It also appears to slow the rates of depolarization and repolarization of muscle cell membranes.
Imidourea Absorption
Rapidly absorbed and distributed
Imidourea side effects and Toxicity
LD50 = 475 mg/kg (oral, rat). Can cause severe gastrointestinal symptoms (nausea, vomiting and diarrhea), bone marrow suppression, renal insufficiency and other hematologic abnormalities (anemia, leucopenia). Severe guanidine intoxication is characterized by nervous hyperirritability, fibrillary tremors and convulsive contractions of muscle, salivation, vomiting, diarrhea, hypoglycemia, and circulatory disturbances.
Imidourea Patient Information
No information avaliable
Imidourea Organisms Affected
Humans and other mammals