Glauco-Visken




Glauco-Visken Brand names, Glauco-Visken Analogs
Glauco-Visken Brand Names Mixture
- Viskazide 10/25tab (Hydrochlorothiazide + Pindolol)
- Viskazide 10/50tab (Hydrochlorothiazide + Pindolol)
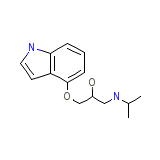
Glauco-Visken Chemical_Formula
C14H20N2O2
Glauco-Visken RX_link
http://www.rxlist.com/cgi/generic3/pindolol.htm
Glauco-Visken fda sheet
Glauco-Visken msds (material safety sheet)
Glauco-Visken Synthesis Reference
No information avaliable
Glauco-Visken Molecular Weight
248.321 g/mol
Glauco-Visken Melting Point
167-171 oC
Glauco-Visken H2O Solubility
7880 mg/L
Glauco-Visken State
Solid
Glauco-Visken LogP
2.525
Glauco-Visken Dosage Forms
Tablet
Glauco-Visken Indication
For the management of hypertension, edema, ventricular tachycardias, and atrial fibrillation.
Glauco-Visken Pharmacology
Pindolol is a non-selective beta-adrenergic antagonist (beta-blocker) which possesses intrinsic sympathomimetic activity (ISA) in therapeutic dosage ranges but does not possess quinidine-like membrane stabilizing activity. Pindolol impairs AV node conduction and decreases sinus rate and may also increase plasma triglycerides and decrease HDL-cholesterol levels. Pindolol is nonpolar and hydrophobic, with low to moderate lipid solubility. Pindolol has little to no intrinsic sympathomimetic activity and, unlike some other beta-adrenergic blocking agents, pindolol has little direct myocardial depressant activity and does not have an anesthetic-like membrane-stabilizing action.
Glauco-Visken Absorption
Rapidly and reproducibly absorbed (bioavailability greater than 95%).
Glauco-Visken side effects and Toxicity
LD50=263 mg/kg (orally in rats). Signs of overdose include excessive bradycardia, cardiac failure, hypotension, and bronchospasm.
Glauco-Visken Patient Information
Glauco-Visken Organisms Affected
Humans and other mammals