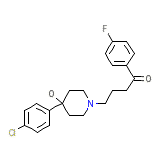
Uliolind




Uliolind Brand names, Uliolind Analogs
- ALDO
- Aloperidin
- Aloperidol
- Aloperidolo
- Aloperidon
- Apo-Haloperidol
- Bioperidolo
- Brotopon
- Dozic
- Dozix
- Einalon S
- Eukystol
- Galoperidol
- Haldol
- Haldol Decanoate
- Haldol La
- Haldol Solutab
- Halidol
- Halojust
- Halol
- Halopal
- Haloperido
- Haloperidol Decanoate
- Haloperidol Intensol
- Haloperidol Lactate
- Halopidol
- Halopoidol
- Halosten
- Keselan
- Lealgin Compositum
- Linton
- Mixidol
- Novo-Peridol
- Pekuces
- Peluces
- Peridol
- Pernox
- Pms Haloperidol
- Serenace
- Serenase
- Serenelfi
- Sernas
- Sernel
- Sigaperidol
- Ulcolind
- Uliolind
- Vesalium
Uliolind Brand Names Mixture
- No information avaliable
Uliolind Chemical_Formula
C21H23ClFNO2
Uliolind RX_link
http://www.rxlist.com/cgi/generic/haloper.htm
Uliolind fda sheet
Uliolind msds (material safety sheet)
Uliolind Synthesis Reference
P. A. J. Janssen, Belg. pat. 577,977 (1959), C.A. 54, 4630c (1960); idem, Brit. pat. 895,309 corresp to U.S. pat. 3,438,991 (1962, 1969 to Janssen).
Uliolind Molecular Weight
375.864 g/mol
Uliolind Melting Point
151.5 oC
Uliolind H2O Solubility
14 mg/L
Uliolind State
Solid
Uliolind LogP
4.362
Uliolind Dosage Forms
Injection; Oral solution; Tablet
Uliolind Indication
For the treatment of schizophrenic patients who require prolonged parenteral antipsychotic therapy also used in Tourette's syndrome and Severe hyperactivity.
Uliolind Pharmacology
Haloperidol is a psychotropic agent indicated for the treatment of schizophrenia. It also exerts sedative and antiemetic activity. Haloperidol has actions at all levels of the central nervous system-primarily at subcortical levels-as well as on multiple organ systems. Haloperidol has strong antiadrenergic and weaker peripheral anticholinergic activity; ganglionic blocking action is relatively slight. It also possesses slight antihistaminic and antiserotonin activity.
Uliolind Absorption
Oral-60%
Uliolind side effects and Toxicity
LD50=165 mg/kg (rats, oral)
Uliolind Patient Information
PRD00538_PI.txt
Uliolind Organisms Affected
Humans and other mammals