Relif




Relif Brand names, Relif Analogs
Relif Brand Names Mixture
- No information avaliable
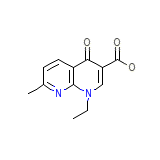
Relif Chemical_Formula
C12H12N2O3
Relif RX_link
http://www.rxlist.com/cgi/generic2/nalidixicacid.htm
Relif fda sheet
Relif msds (material safety sheet)
Relif Synthesis Reference
No information avaliable
Relif Molecular Weight
232.235 g/mol
Relif Melting Point
229.5 oC
Relif H2O Solubility
100 mg/L
Relif State
Solid
Relif LogP
0.949
Relif Dosage Forms
Suspension (250 mg/5 mL tsp); Tablets (1000 mg, 500 mg, 250 mg)
Relif Indication
For the treatment of urinary tract infections caused by susceptible gram-negative microorganisms, including the majority of E. Coli, Enterobacter species, Klebsiella species, and Proteus species.
Relif Pharmacology
Nalidixic acid is a quinolone antibacterial agent for oral administration. Nalidixic acid has marked antibacterial activity against gram-negative bacteria including Enterobacter species, Escherichia coli, Morganella Morganii; Proteus Mirabilis, Proteus vulgaris, and Providencia rettgeri. Pseudomonas species are generally resistant to the drug. Nalidixic acid is bactericidal and is effective over the entire urinary pH range. Conventional chromosomal resistance to nalidixic acid taken in full dosage has been reported to emerge in approximately 2 to 14 percent of patients during treatment; however, bacterial resistance to nalidixic acid has not been shown to be transferable via R factor.
Relif Absorption
Following oral administration, nalidixic acid is rapidly absorbed from the gastrointestinal tract. Bioavailability is approximately 96%. Absorption may be delayed if taken with antacids.
Relif side effects and Toxicity
ORAL (LD50): Acute: 1160 mg/kg [Rat]. 572 mg/kg [Mouse]. Toxic psychosis, convulsions, increased intracranial pressure, or metabolic acidosis may occur in patients taking more than the recommended dosage. Vomiting, nausea, and lethargy may also occur following overdosage.
Relif Patient Information
Relif Organisms Affected
Enteric bacteria and other eubacteria