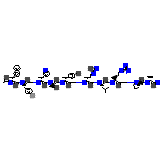
Pyrisept




Pyrisept Brand names, Pyrisept Analogs
Pyrisept Brand Names Mixture
- No information avaliable
Pyrisept Chemical_Formula
No information avaliable
Pyrisept RX_link
http://www.rxlist.com/cgi/generic2/cetrorelix.htm
Pyrisept fda sheet
Pyrisept msds (material safety sheet)
Pyrisept Synthesis Reference
No information avaliable
Pyrisept Molecular Weight
1431.06 g/mol
Pyrisept Melting Point
No information avaliable
Pyrisept H2O Solubility
No information avaliable
Pyrisept State
Solid
Pyrisept LogP
No information avaliable
Pyrisept Dosage Forms
Powder for solution
Pyrisept Indication
For the inhibition of premature LH surges in women undergoing controlled ovarian stimulation.
Pyrisept Pharmacology
Cetrorelix is a synthetic decapeptide with gonadotropin-releasing hormone (GnRH) antagonistic activity. GnRH induces the production and release of luteinizing hormone (LH) and follicle stimulating hormone (FSH) from the gonadotrophic cells of the anterior pituitary. Due to a positive estradiol (E2) feedback at midcycle, GnRH liberation is enhanced resulting in an LH-surge. This LH-surge induces the ovulation of the dominant follicle, resumption of oocyte meiosis and subsequently luteinization as indicated by rising progesterone levels. Cetrorelix competes with natural GnRH for binding to membrane receptors on pituitary cells and thus controls the release of LH and FSH in a dose-dependent manner.
Pyrisept Absorption
Rapidly absorbed following subcutaneous injection. The mean absolute bioavailability following subcutaneous administration to healthy female subjects is 85%.
Pyrisept side effects and Toxicity
No information avaliable
Pyrisept Patient Information
Pyrisept Organisms Affected
Humans and other mammals