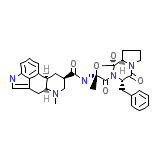
DHE-45




DHE-45 Brand names, DHE-45 Analogs
- Agit
- Angionorm
- D.H.E.
- D.H.E. 45
- DET MS
- DHE-45
- Dergotamine
- Diergo
- Dihidroergotamina [INN-Spanish]
- Dihydergot
- Dihydroergotamine mesylate
- Dihydroergotamine methanesulfonate
- Dihydroergotamine monomethanesulfonate
- Dihydroergotaminum [INN-Latin]
- Dirgotarl
- Endophleban
- Ergomimet
- Ergont
- Ergotamine, 9,10-dihydro-
- Ergotonin
- Ikaran
- Migranal
- Morena
- Orstanorm
- Tonopres
- Verladyn
DHE-45 Brand Names Mixture
- No information avaliable
DHE-45 Chemical_Formula
C33H37N5O5
DHE-45 RX_link
http://www.rxlist.com/cgi/generic2/dihyergmes.htm
DHE-45 fda sheet
DHE-45 msds (material safety sheet)
DHE-45 Synthesis Reference
No information avaliable
DHE-45 Molecular Weight
583.678 g/mol
DHE-45 Melting Point
No information avaliable
DHE-45 H2O Solubility
No information avaliable
DHE-45 State
Solid
DHE-45 LogP
No information avaliable
DHE-45 Dosage Forms
Liquid
DHE-45 Indication
For the acute treatment of migraine headaches with or without aura and the acute treatment of cluster headache episodes.
DHE-45 Pharmacology
Dihydroergotamine is indicated for the acute treatment of migraine headaches with or without aura and the acute treatment of cluster headache episodes. Dihydroergotamine binds with high affinity to 5-HT1Da and 5-HT1Db receptors. It also binds with high affinity to serotonin 5-HT1A, 5-HT2A, and 5-HT2C receptors, noradrenaline a2A, a2B and a receptors, and dopamine D2L and D3 receptors. The therapeutic activity of Dihydroergotamine in migraine is generally attributed to the agonist effect at 5-HT1D receptors.
DHE-45 Absorption
Interpatient variable and may be dependent on the administration technique
DHE-45 side effects and Toxicity
Side effects include abdominal pain, abnormal speech, coma, confusion, convulsions, hallucinations, increase and/or decrease in blood pressure, nausea, numbness, tingling, pain, and a bluish color of your fingersand toes, slowed breathing, vomiting
DHE-45 Patient Information
PATIENT INFORMATIOn
Follow the link below to get detailed information before taking D.H.E. 45� (dihydroergotamine mesylate) Injection.
http://www.rxlist.com/cgi/generic2/dihyergmes_pi.htm
Follow the link below to get detailed information before taking D.H.E. 45� (dihydroergotamine mesylate) Injection.
http://www.rxlist.com/cgi/generic2/dihyergmes_pi.htm
DHE-45 Organisms Affected
Humans and other mammals