Endophleban




Endophleban Brand names, Endophleban Analogs
- Agit
- Angionorm
- D.H.E.
- D.H.E. 45
- DET MS
- DHE-45
- Dergotamine
- Diergo
- Dihidroergotamina [INN-Spanish]
- Dihydergot
- Dihydroergotamine mesylate
- Dihydroergotamine methanesulfonate
- Dihydroergotamine monomethanesulfonate
- Dihydroergotaminum [INN-Latin]
- Dirgotarl
- Endophleban
- Ergomimet
- Ergont
- Ergotamine, 9,10-dihydro-
- Ergotonin
- Ikaran
- Migranal
- Morena
- Orstanorm
- Tonopres
- Verladyn
Endophleban Brand Names Mixture
- No information avaliable
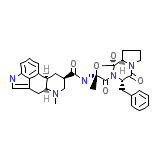
Endophleban Chemical_Formula
C33H37N5O5
Endophleban RX_link
http://www.rxlist.com/cgi/generic2/dihyergmes.htm
Endophleban fda sheet
Endophleban msds (material safety sheet)
Endophleban Synthesis Reference
No information avaliable
Endophleban Molecular Weight
583.678 g/mol
Endophleban Melting Point
No information avaliable
Endophleban H2O Solubility
No information avaliable
Endophleban State
Solid
Endophleban LogP
No information avaliable
Endophleban Dosage Forms
Liquid
Endophleban Indication
For the acute treatment of migraine headaches with or without aura and the acute treatment of cluster headache episodes.
Endophleban Pharmacology
Dihydroergotamine is indicated for the acute treatment of migraine headaches with or without aura and the acute treatment of cluster headache episodes. Dihydroergotamine binds with high affinity to 5-HT1Da and 5-HT1Db receptors. It also binds with high affinity to serotonin 5-HT1A, 5-HT2A, and 5-HT2C receptors, noradrenaline a2A, a2B and a receptors, and dopamine D2L and D3 receptors. The therapeutic activity of Dihydroergotamine in migraine is generally attributed to the agonist effect at 5-HT1D receptors.
Endophleban Absorption
Interpatient variable and may be dependent on the administration technique
Endophleban side effects and Toxicity
Side effects include abdominal pain, abnormal speech, coma, confusion, convulsions, hallucinations, increase and/or decrease in blood pressure, nausea, numbness, tingling, pain, and a bluish color of your fingersand toes, slowed breathing, vomiting
Endophleban Patient Information
PATIENT INFORMATIOn
Follow the link below to get detailed information before taking D.H.E. 45� (dihydroergotamine mesylate) Injection.
http://www.rxlist.com/cgi/generic2/dihyergmes_pi.htm
Follow the link below to get detailed information before taking D.H.E. 45� (dihydroergotamine mesylate) Injection.
http://www.rxlist.com/cgi/generic2/dihyergmes_pi.htm
Endophleban Organisms Affected
Humans and other mammals