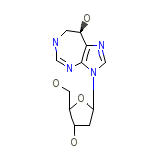
Covidarabine




Covidarabine Brand names, Covidarabine Analogs
Covidarabine Brand Names Mixture
- No information avaliable
Covidarabine Chemical_Formula
C11H16N4O4
Covidarabine RX_link
No information avaliable
Covidarabine fda sheet
Covidarabine msds (material safety sheet)
Covidarabine Synthesis Reference
Showalter, HD et al., J. Med. Chem. 26:1478 (1983)
Covidarabine Molecular Weight
268.269 g/mol
Covidarabine Melting Point
220 oC
Covidarabine H2O Solubility
30 mg/mL
Covidarabine State
Solid
Covidarabine LogP
-1.58
Covidarabine Dosage Forms
Powder for solution
Covidarabine Indication
For the treatment of hairy cell leukaemia refractory to alpha interferon.
Covidarabine Pharmacology
Pentostatin is an antineoplastic anti-metabolite used in the treatment of several forms of leukemia including acute nonlymphocytic leukemia and hairy cell leukemia. Anti-metabolites masquerade as purine or pyrimidine - which become the building blocks of DNA. They prevent these substances becoming incorporated in to DNA during the "S" phase (of the cell cycle), stopping normal development and division. It is a 6-thiopurine analogue of the naturally occurring purine bases hypoxanthine and guanine. Intracellular activation results in incorporation into DNA as a false purine base. An additional cytotoxic effect is related to its incorporation into RNA. Cytotoxicity is cell cycle phase-specific (S-phase).
Covidarabine Absorption
Not absorbed orally, crosses blood brain barrier.
Covidarabine side effects and Toxicity
LD50=128 mg/kg (mouse), side effects include lethargy, rash, fatigue, nausea and myelosuppression.
Covidarabine Patient Information
Covidarabine Organisms Affected
Humans and other mammals