Alendros




Alendros Brand names, Alendros Analogs
Alendros Brand Names Mixture
- No information avaliable
Alendros Chemical_Formula
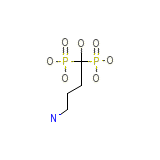
C4H13NO7P2
Alendros RX_link
http://www.rxlist.com/cgi/generic/alendron.htm
Alendros fda sheet
Alendros msds (material safety sheet)
Alendros Synthesis Reference
M. I. Kabachnik et al., Bull. Acad. Sci. USSR,.27, 374 (1978)
Alendros Molecular Weight
249.096 g/mol
Alendros Melting Point
233 - 235 oC
Alendros H2O Solubility
1mg/L
Alendros State
Solid
Alendros LogP
-3.198
Alendros Dosage Forms
Tablets (6.53, 13.05, 45.68, 52.21 or 91.37 mg of alendronate monosodium salt trihydrate)
Alendros Indication
For the treatment and prevention of osteoporosis in women and Paget's disease of bone in both men and women.
Alendros Pharmacology
Alendronate, a second-generation bisphosphonate is the first member of a group of drugs which strengthens bone. Alendronate is used to reduce hypercalcemia in tumor-induced bone disease, to treat corticosteroid-induced osteoporosis and Paget's disease, and to prevent osteoporosis in postmenopausal women.
Alendros Absorption
Relative to an intravenous (IV) reference dose, the mean oral bioavailability of alendronate in women was 0.7% for doses ranging from 5 to 40 mg when administered after an overnight fast and two hours before a standardized breakfast. Oral bioavailability of the 10 mg tablet in men (0.59%) was similar to that in women (0.78%) when administered after an overnight fast and 2 hours before breakfast.
Alendros side effects and Toxicity
Alendronate can damage the esophagus both by toxicity from the medication itself and by nonspecific irritation secondary to contact between the pill and the esophageal mucosa, similar to other cases of "pill esophagitis."
Alendros Patient Information
Alendros Organisms Affected
Humans and other mammals