Yutopar S.R




Yutopar S.R Brand names, Yutopar S.R Analogs
Yutopar S.R Brand Names Mixture
- No information avaliable
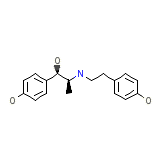
Yutopar S.R Chemical_Formula
C17H21NO3
Yutopar S.R RX_link
No information avaliable
Yutopar S.R fda sheet
Yutopar S.R msds (material safety sheet)
Yutopar S.R Synthesis Reference
Belg. pat. 660.244 (1965 to N.V. Philips), C.A. 63, 17965h (1965) corresp to Claassen et al., U.S. pat. 3,410,944 (1968 to No. Am. Philips)
Yutopar S.R Molecular Weight
287.354 g/mol
Yutopar S.R Melting Point
88-90 oC
Yutopar S.R H2O Solubility
Complete
Yutopar S.R State
Solid
Yutopar S.R LogP
2.588
Yutopar S.R Dosage Forms
Solution for IV; Capsule (extended-release); Tablet; Intramuscular injection
Yutopar S.R Indication
For the treatment and prophylaxis of premature labour
Yutopar S.R Pharmacology
Beta-2 adrenergic receptors are located at sympathetic neuroeffector junctions of many organs, including uterus. Ritodrine is beta-2 adrenergic agonist. It stimulates beta-2 adrenergic receptor, increases cAMP level and decreases intracellular calcium concentration. The decrease of calcium concentration leads to a relaxation of uterine smooth muscle.
Yutopar S.R Absorption
No information avaliable
Yutopar S.R side effects and Toxicity
LD50=64mg/kg (mice, IV); LD50=540 mg/kg (mice, oral); LD50=85 mg/kg (rat, IV)
Yutopar S.R Patient Information
Yutopar S.R Organisms Affected
Humans and other mammals