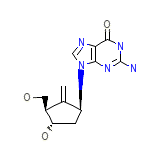
Palavale




Palavale Brand names, Palavale Analogs
Palavale Brand Names Mixture
- No information avaliable
Palavale Chemical_Formula
C12H15N5O3
Palavale RX_link
http://www.rxlist.com/cgi/generic4/baraclude.htm
Palavale fda sheet
Palavale msds (material safety sheet)
Palavale Synthesis Reference
No information avaliable
Palavale Molecular Weight
277.279 g/mol
Palavale Melting Point
No information avaliable
Palavale H2O Solubility
Slightly soluble (2.4 mg/mL at pH 7.9, 25oC)
Palavale State
Solid
Palavale LogP
No information avaliable
Palavale Dosage Forms
Film-coated tablets for oral administration (0.5 mg, 1 mg); Oral solution (0.05 mg/mL)
Palavale Indication
For the treatment of chronic hepatitis B virus infection in adults with evidence of active viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease.
Palavale Pharmacology
Entecavir is a guanosine nucleoside analogue with selective activity against hepatitis B virus (HBV). It is designed to selectively inhibit the Hepatitis B virus, blocking all three steps in the replication process. Entecavir is more efficient than an older Hepatitis B drug, lamivudine.
Palavale Absorption
Absorption Following oral administration in healthy subjects, entecavir peak plasma concentrations occurred between 0.5 and 1.5 hours. In healthy subjects, the bioavailability of the tablet is 100% relative to the oral solution.
Palavale side effects and Toxicity
No information avaliable
Palavale Patient Information
Palavale Organisms Affected
Hepatitis B