Mepivicaine




Mepivicaine Brand names, Mepivicaine Analogs
Mepivicaine Brand Names Mixture
- No information avaliable
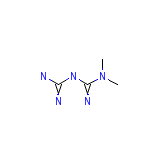
Mepivicaine Chemical_Formula
C4H11N5
Mepivicaine RX_link
http://www.rxlist.com/cgi/generic4/glumetza.htm
Mepivicaine fda sheet
Mepivicaine msds (material safety sheet)
Mepivicaine Synthesis Reference
No information avaliable
Mepivicaine Molecular Weight
129.164 g/mol
Mepivicaine Melting Point
223-226 oC
Mepivicaine H2O Solubility
Freely soluble as HCl salt
Mepivicaine State
Solid
Mepivicaine LogP
-0.267
Mepivicaine Dosage Forms
Extended release tablet (500 mg or 1000 mg)
Mepivicaine Indication
For use as an adjunct to diet and exercise to improve glycemic control in adult patients (18 years and older) with type 2 diabetes.
Mepivicaine Pharmacology
Metformin is an antihyperglycemic agent, which improves glucose tolerance in patients with type 2 diabetes, lowering both basal and postprandial plasma glucose. Metformin is not chemically or pharmacologically related to any other classes of oral antihyperglycemic agents. Unlike sulfonylureas, metformin does not produce hypoglycemia in either patients with type 2 diabetes or normal subjects and does not cause hyperinsulinemia. With metformin therapy, insulin secretion remains unchanged while fasting insulin levels and daylong plasma insulin response may actually decrease.
Mepivicaine Absorption
Absorbed over 6 hours, bioavailability is 50 to 60% under fasting conditions. Food delays absorption.
Mepivicaine side effects and Toxicity
Acute oral toxicity (LD50): 350 mg/kg [Rabbit]. It would be expected that adverse reactions of a more intense character including epigastric discomfort, nausea, and vomiting followed by diarrhea, drowsiness, weakness, dizziness, malaise and headache might be seen.
Mepivicaine Patient Information
Mepivicaine Organisms Affected
Humans and other mammals