EN 1530 Base




EN 1530 Base Brand names, EN 1530 Base Analogs
EN 1530 Base Brand Names Mixture
- No information avaliable
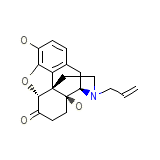
EN 1530 Base Chemical_Formula
C19H21NO4
EN 1530 Base RX_link
http://www.rxlist.com/cgi/generic3/naloxone.htm
EN 1530 Base fda sheet
EN 1530 Base msds (material safety sheet)
EN 1530 Base Synthesis Reference
No information avaliable
EN 1530 Base Molecular Weight
327.374 g/mol
EN 1530 Base Melting Point
200 - 205 oC
EN 1530 Base H2O Solubility
Soluble
EN 1530 Base State
Solid
EN 1530 Base LogP
1.464
EN 1530 Base Dosage Forms
Liquid
EN 1530 Base Indication
For the complete or partial reversal of narcotic depression, including respiratory depression, induced by opioids including natural and synthetic narcotics, propoxyphene, methadone and the narcotic-antagonist analgesics: nalbuphine, pentazocine and butorphanol.
EN 1530 Base Pharmacology
Naloxone is an opiate antagonist and prevents or reverses the effects of opioids including respiratory depression, sedation and hypotension. Also, it can reverse the psychotomimetic and dysphoric effects of agonist-antagonists such as pentazocine. Naloxone is an essentially pure narcotic antagonist, i.e., it does not possess the "agonistic" or morphine-like properties characteristic of other narcotic antagonists; naloxone does not produce respiratory depression, psychotomimetic effects or pupillary constriction. In the absence of narcotics or agonistic effects of other narcotic antagonists, it exhibits essentially no pharmacologic activity.
EN 1530 Base Absorption
Well absorbed following intramuscular injection.
EN 1530 Base side effects and Toxicity
No information avaliable
EN 1530 Base Patient Information
No information avaliable
EN 1530 Base Organisms Affected
Humans and other mammals