Solantoin




Solantoin Brand names, Solantoin Analogs
- 5,5-Dwufenylohydantoina
- 5,5-Dwufenylohydantoina [Polish]
- Aleviatin
- Antisacer
- Auranile
- Causoin
- Cerebyx
- Citrullamon
- Citrulliamon
- Comital
- Comitoina
- Convul
- DPH
- DPH (VAN)
- Danten
- Dantinal
- Dantoinal
- Dantoinal klinos
- Dantoine
- Denyl
- Di-Hydan
- Di-Lan
- Di-Lan (VAN)
- Di-Phetine
- Didan TDC 250
- Didan-tdc-250
- Difenilhidantoina
- Difenilhidantoina (spanish)
- Difenilhidantoina [Spanish]
- Difenin
- Difetoin
- Difhydan
- Dihycon
- Dihydantoin
- Dilabid
- Dilantin
- Dilantin acid
- Dilantin-125
- Dilantine
- Dillantin
- Dintoin
- Dintoina
- Diphantoin
- Diphedal
- Diphedan
- Diphenat
- Diphenin
- Diphenine
- Diphentoin
- Diphentyn
- Diphenylan
- Diphenylan Sodium
- Diphenylhydantoin
- Diphenylhydantoin (VAN)
- Diphenylhydantoine
- Diphenylhydantoine [French]
- Diphenylhydatanoin
- Ditoinate
- Ekko
- Ekko capsules
- Elepsindon
- Enkelfel
- Epamin
- Epanutin
- Epasmir "5"
- Epasmir 5
- Epdantoin Simple
- Epdantoine simple
- Epelin
- Epifenyl
- Epihydan
- Epilan
- Epilan D
- Epilan-D
- Epilantin
- Epinat
- Epised
- Eptal
- Eptoin
- Extended Phenytoin Sodium
- Fenantoin
- Fenantoin Mn Pharma
- Fenidantoin "s"
- Fenidantoin s
- Fenitoina
- Fenitoina [INN-Spanish]
- Fentoin
- Fenylepsin
- Fenytoin Dak
- Fenytoine
- Fosphenytoin
- Gerot-epilan-D
- Hidan
- Hidantal
- Hidantilo
- Hidantina
- Hidantina senosian
- Hidantina vitoria
- Hidantomin
- Hindatal
- Hydantal
- Hydantin
- Hydantoin
- Hydantoinal
- Hydantol
- Ictalis simple
- Idantoil
- Idantoin
- Iphenylhydantoin
- Kessodanten
- Labopal
- Lehydan
- Lepitoin
- Lepsin
- Mesantoin
- Minetoin
- Neos-Hidantoina
- Neosidantoina
- Novantoina
- Novophenytoin
- Om hidantoina simple
- Om-Hydantoine
- Oxylan
- Phanantin
- Phanatine
- Phenatine
- Phenatoine
- Phenhydan
- Phenhydanin
- Phenitoin
- Phentoin
- Phentytoin
- Phenytex
- Phenytoin AWD
- Phenytoin Base
- Phenytoin Sodium
- Phenytoin USP
- Phenytoin-Gerot
- Phenytoine
- Phenytoine [INN-French]
- Phenytoinum [INN-Latin]
- Prompt Phenytoin Sodium
- Ritmenal
- Saceril
- Sanepil
- Silantin
- Sinergina
- Sodanthon
- Sodantoin
- Sodanton
- Solantin
- Solantoin
- Solantyl
- Sylantoic
- TOIN
- Tacosal
- Thilophenyl
- Toin unicelles
- Zentronal
- Zentropil
Solantoin Brand Names Mixture
- Dilantin W Phenobarbital 15mg (Phenobarbital + Phenytoin Sodium)
- Dilantin W Phenobarbital 30mg Cap (Phenobarbital + Phenytoin Sodium)
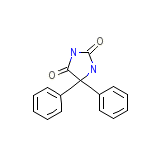
Solantoin Chemical_Formula
C15H12N2O2
Solantoin RX_link
http://www.rxlist.com/cgi/generic/phenyt.htm
Solantoin fda sheet
Solantoin msds (material safety sheet)
Solantoin Synthesis Reference
Henze, U.S. Pat. 2,409,754 (1946)
Solantoin Molecular Weight
252.268 g/mol
Solantoin Melting Point
286 oC
Solantoin H2O Solubility
32 mg/L
Solantoin State
Solid
Solantoin LogP
2.241
Solantoin Dosage Forms
Capsule (extended release); Liquid; Solution; Suspension; Tablet
Solantoin Indication
For the control of generalized tonic-clonic (grand mal) and complex partial (psychomotor, temporal lobe) seizures and prevention and treatment of seizures occurring during or following neurosurgery.
Solantoin Pharmacology
Phenytoin is an antiepileptic drug which can be useful in the treatment of epilepsy. The primary site of action appears to be the motor cortex where spread of seizure activity is inhibited. Phenytoin reduces the maximal activity of brain stem centers responsible for the tonic phase of tonic-clonic (grand mal) seizures. Phenytoin acts to damp the unwanted, runaway brain activity seen in seizure by reducing electrical conductance among brain cells. It lacks the sedation effects associated with phenobarbital. There are some indications that phenytoin has other effects, including anxiety control and mood stabilization, although it has never been approved for those purposes by the FDA.
Solantoin Absorption
Bioavailability 70-100% oral, 24.4% for rectal and intravenous administration. Rapid rate of absorption with peak blood concentration expected in 1½ to 3 hours.
Solantoin side effects and Toxicity
Oral, mouse: LD50 = 150 mg/kg; Oral, rat: LD50 = 1635 mg/kg. Symptoms of overdose include coma, difficulty in pronouncing words correctly, involuntary eye movement, lack of muscle coordination, low blood pressure, nausea, sluggishness, slurred speech, tremors, and vomiting.
Solantoin Patient Information
Patients taking phenytoin should be advised of the importance of adhering strictly to the prescribed dosage regimen, and of informing the physician of any clinical condition in which it is not possible to take the drug orally as prescribed, eg, surgery, etc.
Patients should also be cautioned on the use of other drugs or alcoholic beverages without first seeking the physician's advice.
Patients should be instructed to call their physician if skin rash develops.
The importance of good dental hygiene should be stressed in order to minimize the development of gingival hyperplasia and its complications.
Do not use capsules which are discolored.
Solantoin Organisms Affected
Humans and other mammals