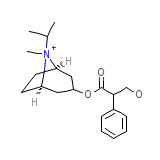
Rinovagos




Rinovagos Brand names, Rinovagos Analogs
Rinovagos Brand Names Mixture
- Combivent Inhalation Aerosol (Ipratropium Bromide + Salbutamol Sulfate)
- Combivent Inhalation Solution (Ipratropium Bromide + Salbutamol Sulfate)
- Duovent Udv (Fenoterol Hydrobromide + Ipratropium Bromide)
- Ratio-Ipra Sal Udv (Ipratropium Bromide + Salbutamol Sulfate)
Rinovagos Chemical_Formula
C20H30NO3
Rinovagos RX_link
http://www.rxlist.com/cgi/generic/ipratrop.htm
Rinovagos fda sheet
Rinovagos msds (material safety sheet)
Rinovagos Synthesis Reference
K. Zeile et al.,U.S. Pat. 3,505,337 (1970)
Rinovagos Molecular Weight
332.457 g/mol
Rinovagos Melting Point
230-232oC
Rinovagos H2O Solubility
Freely soluble
Rinovagos State
Solid
Rinovagos LogP
No information avaliable
Rinovagos Dosage Forms
Aerosol (with propellants); Liquid; Metered-dose (aerosol); Metered-dose (pump); Solution
Rinovagos Indication
For maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease, including chronic bronchitis and emphysema.
Rinovagos Pharmacology
Ipratropium bromide, a synthetic ammonium compound structurally similar to atropine, is used as a bronchodilator in the management of cholinergic-mediated bronchospasm associated with chronic obstructive pulmonary disease and in the treatment of rhinorrhea associated with the common cold or with allergic or nonallergic seasonal rhinitis.
Rinovagos Absorption
Inhalation (local)-minimal; Nasal-rapid and minimal
Rinovagos side effects and Toxicity
LD50=1001mg/kg (orally in mice)
Rinovagos Patient Information
Rinovagos Organisms Affected
Humans and other mammals