Lidanar




Lidanar Brand names, Lidanar Analogs
Lidanar Brand Names Mixture
- No information avaliable
Lidanar Chemical_Formula
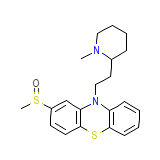
C21H26N2OS2
Lidanar RX_link
http://www.rxlist.com/cgi/generic3/mesoridazine.htm
Lidanar fda sheet
Lidanar msds (material safety sheet)
Lidanar Synthesis Reference
No information avaliable
Lidanar Molecular Weight
386.576 g/mol
Lidanar Melting Point
No information avaliable
Lidanar H2O Solubility
No information avaliable
Lidanar State
Solid
Lidanar LogP
4.485
Lidanar Dosage Forms
Tablet (10 mg, 25 mg, 50 mg, 100 mg) for oral administration; Ampuls (1 mL) for intramuscular administration; Concentrate for oral administration
Lidanar Indication
Used in the treatment of schizophrenia, organic brain disorders, alcoholism and psychoneuroses.
Lidanar Pharmacology
Mesoridazine, the besylate salt of a metabolite of thioridazine, is a phenothiazine tranquilizer. Pharmacological studies in laboratory animals have established that mesoridazine has a spectrum of pharmacodynamic actions typical of a major tranquilizer. In common with other tranquilizers it inhibits spontaneous motor activity in mice, prolongs thiopental and hexobarbital sleeping time in mice and produces spindles and block of arousal reaction in the EEG of rabbits. It is effective in blocking spinal reflexes in the cut and antagonizes d-amphetamine excitation and toxicity in grouped mice. It shows a moderate adrenergic blocking activity in vitro and in vivo and antagonizes 5-hydroxytryptamine in vivo. Intravenously administered, it lowers the blood pressure of anesthetized dogs. It has a weak antiacetylcholine effect in vitro.
Lidanar Absorption
Well absorbed from the gastrointestinal tract.
Lidanar side effects and Toxicity
Oral LD50 is 560 ± 62.5 mg/kg and 644 ± 48 mg/kg in mouse and rat, respectively. Symptoms of overdose may include emesis, muscle tremors, decreased food intake and death associated with aspiration of oral-gastric contents into the respiratory system.
Lidanar Patient Information
Lidanar Organisms Affected
Humans and other mammals