Dea No. 2884




Dea No. 2884 Brand names, Dea No. 2884 Analogs
Dea No. 2884 Brand Names Mixture
- No information avaliable
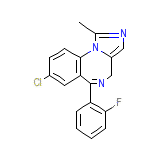
Dea No. 2884 Chemical_Formula
C18H13ClFN3
Dea No. 2884 RX_link
http://www.rxlist.com/cgi/generic2/versedsyr.htm
Dea No. 2884 fda sheet
Dea No. 2884 msds (material safety sheet)
Dea No. 2884 Synthesis Reference
No information avaliable
Dea No. 2884 Molecular Weight
325.767 g/mol
Dea No. 2884 Melting Point
159 oC
Dea No. 2884 H2O Solubility
40.0 mg/ml
Dea No. 2884 State
Solid
Dea No. 2884 LogP
3.868
Dea No. 2884 Dosage Forms
Liquid; Solution
Dea No. 2884 Indication
For use in pediatric patients for sedation, anxiolysis, and amnesia prior to diagnostic, therapeutic, or endoscopic procedures or before induction of anesthesia.
Dea No. 2884 Pharmacology
Midazolam is a short-acting benzodiazepine central nervous system (CNS) depressant. Pharmacodynamic properties of midazolam and its metabolites, which are similar to those of other benzodiazepines, include sedative, anxiolytic, amnesic and hypnotic activities. Benzodiazepine pharmacologic effects appear to result from reversible interactions with the (gamma)-amino butyric acid (GABA) benzodiazepine receptor in the CNS, the major inhibitory neurotransmitter in the central nervous system. The action of midazolam is readily reversed by the benzodiazepine receptor antagonist, flumazenil.
Dea No. 2884 Absorption
Rapidly absorbed after oral administration (absolute bioavailability of the midazolam syrup in pediatric patients is about 36%, and intramuscular is greater than 90%).
Dea No. 2884 side effects and Toxicity
LD50=825 mg/kg (Orally in rats). Signs of overdose include sedation, somnolence, confusion, impaired coordination, diminished reflexes, coma, and deleterious effects on vital signs.
Dea No. 2884 Patient Information
Dea No. 2884 Organisms Affected
Humans and other mammals