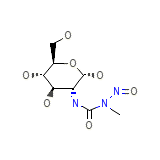
Zanosar




Zanosar Brand names, Zanosar Analogs
Zanosar Brand Names Mixture
- No information avaliable
Zanosar Chemical_Formula
C8H15N3O7
Zanosar RX_link
http://www.rxlist.com/cgi/generic/zanosar.htm
Zanosar fda sheet
Zanosar msds (material safety sheet)
Zanosar Synthesis Reference
No information avaliable
Zanosar Molecular Weight
265.221 g/mol
Zanosar Melting Point
115 oC
Zanosar H2O Solubility
5070 mg/L
Zanosar State
Solid
Zanosar LogP
-2.958
Zanosar Dosage Forms
Powder for solution
Zanosar Indication
For the treatment of malignant neoplasms of pancreas (metastatic islet cell carcinoma).
Zanosar Pharmacology
Streptozocin is an antitumour antibiotic consisting of a nitrosourea moiety interposed between a methyl group and a glucosamine. Streptozocin is indicated in the treatment of metastatic islet cell carcinoma of the pancreas. Streptozocin inhibits DNA synthesis in bacterial and mammalian cells. In bacterial cells, a specific interaction with cytosine moieties leads to degradation of DNA. The biochemical mechanism leading to mammalian cell death has not been definitely established; streptozocin inhibits cell proliferation at a considerably lower level than that needed to inhibit precursor incorporation into DNA or to inhibit several of the enzymes involved in DNA synthesis. Although streptozocin inhibits the progression of cells into mitosis, no specific phase of the cell cycle is particularly sensitive to its lethal effects.
Zanosar Absorption
Poor oral absorption (17-25%)
Zanosar side effects and Toxicity
Symptoms of overdose include nausea and vomiting, anorexia, myelosuppression; and nephrotoxicity.
Zanosar Patient Information
No information avaliable
Zanosar Organisms Affected
Humans and other mammals