VDN




VDN Brand names, VDN Analogs
VDN Brand Names Mixture
- No information avaliable
VDN Chemical_Formula
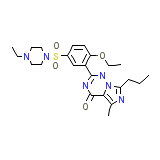
C23H32N6O4S
VDN RX_link
http://www.rxlist.com/cgi/generic3/levitra.htm
VDN fda sheet
VDN msds (material safety sheet)
VDN Synthesis Reference
No information avaliable
VDN Molecular Weight
488.604 g/mol
VDN Melting Point
192 oC
VDN H2O Solubility
0.11 mg/mL (HCl salt)
VDN State
Solid
VDN LogP
No information avaliable
VDN Dosage Forms
Tablet (5, 10, and 20 mg)
VDN Indication
Used for the treatment of erectile dysfunction
VDN Pharmacology
Vardenafil is used to treat male erectile dysfunction (impotence) and pulmonary arterial hypertension (PAH). Part of the physiological process of erection involves the release of nitric oxide (NO) in the corpus cavernosum. This then activates the enzyme guanylate cyclase which results in increased levels of cyclic guanosine monophosphate (cGMP), leading to smooth muscle relaxation in the corpus cavernosum, resulting in increased inflow of blood and an erection. Vardenafil is a potent and selective inhibitor of cGMP specific phosphodiesterase type 5 (PDE5) which is responsible for degradation of cGMP in the corpus cavernosum. This means that, with vardenafil on board, normal sexual stimulation leads to increased levels of cGMP in the corpus cavernosum which leads to better erections. Without sexual stimulation and no activation of the NO/cGMP system, vardenafil should not cause an erection.
VDN Absorption
Vardenafil is rapidly absorbed with absolute bioavailability of approximately 15%.
VDN side effects and Toxicity
Symptoms of overdose include vision changes and back and muscle pain.
VDN Patient Information
VDN Organisms Affected
Humans and other mammals