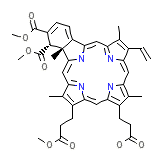
Trifluorothymidine




Trifluorothymidine Brand names, Trifluorothymidine Analogs
Trifluorothymidine Brand Names Mixture
- No information avaliable
Trifluorothymidine Chemical_Formula
C41H42N4O8
Trifluorothymidine RX_link
http://www.rxlist.com/cgi/generic2/verteporfin.htm
Trifluorothymidine fda sheet
Trifluorothymidine msds (material safety sheet)
Trifluorothymidine Synthesis Reference
No information avaliable
Trifluorothymidine Molecular Weight
718.794 g/mol
Trifluorothymidine Melting Point
No information avaliable
Trifluorothymidine H2O Solubility
No information avaliable
Trifluorothymidine State
Solid
Trifluorothymidine LogP
3.743
Trifluorothymidine Dosage Forms
Powder for solution to be mixed for injection (2mg verteporfin/mL reconstituted verteporfin)
Trifluorothymidine Indication
For the treatment of patients with predominantly classic subfoveal choroidal neovascularization due to age-related macular degeneration, pathologic myopia or presumed ocular histoplasmosis.
Trifluorothymidine Pharmacology
Verteporfin, otherwise known as benzoporphyrin derivative, is a medication used in conjunction with laser treatment to eliminate the abnormal blood vessels in the eye associated with conditions such as the wet form of macular degeneration. Verteporfin accumulates in these abnormal blood vessels and, when stimulated by nonthermal red light with a wavelength of 693 nm in the presence of oxygen, produces highly reactive short-lived singlet oxygen and other reactive oxygen radicals, resulting in local damage to the endothelium and blockage of the vessels.
Trifluorothymidine Absorption
No information avaliable
Trifluorothymidine side effects and Toxicity
Overdose of drug and/or light in the treated eye may result in nonperfusion of normal retinal vessels with the possibility of severe decrease in vision that could be permanent. An overdose of drug will also result in the prolongation of the period during which the patient remains photosensitive to bright light.
Trifluorothymidine Patient Information
Trifluorothymidine Organisms Affected
Humans and other mammals