Protonix IV




Protonix IV Brand names, Protonix IV Analogs
Protonix IV Brand Names Mixture
- No information avaliable
Protonix IV Chemical_Formula
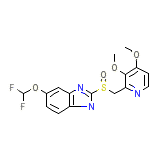
C16H15F2N3O4S
Protonix IV RX_link
http://www.rxlist.com/cgi/generic3/protonix.htm
Protonix IV fda sheet
Protonix IV msds (material safety sheet)
Protonix IV Synthesis Reference
No information avaliable
Protonix IV Molecular Weight
383.371 g/mol
Protonix IV Melting Point
Because of gradual degradation of pantoprazole sodium during heating, the melting point cannot be determined.
Protonix IV H2O Solubility
Freely soluble in water.
Protonix IV State
Solid
Protonix IV LogP
1.268
Protonix IV Dosage Forms
Tablet (enteric-coated)
Protonix IV Indication
Short-term (up to 16 weeks) treatment of erosive esophagitis.
Protonix IV Pharmacology
Pantoprazole is a substituted benzimidazole indicated for the short-term treatment (up to 16 weeks) in the healing and symptomatic relief of erosive esophagitis. Pantoprazole is a proton pump inhibitor (PPI) that suppresses the final step in gastric acid production.
Protonix IV Absorption
Pantoprazole is well absorbed; it undergoes little first-pass metabolism resulting in an absolute bioavailability of approximately 77%.
Protonix IV side effects and Toxicity
No information avaliable
Protonix IV Patient Information
Patients should be cautioned that PROTONIX Delayed-Release Tablets should not be split, crushed or chewed. The tablets should be swallowed whole, with or without food in the stomach. Concomitant administration of antacids does not affect the absorption of pantoprazole.
Protonix IV Organisms Affected
Humans and other mammals