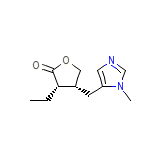
Pilocarpine monohydrochloride




Pilocarpine monohydrochloride Brand names, Pilocarpine monohydrochloride Analogs
- Adsorbocarpine
- Akarpine
- Almocarpine
- Ami-Pilo
- Amistura P
- Beta-pilocarpine hydrochloride
- Epicar
- Isopto Carpine
- Isoptocarpine
- Mi-Pilo
- Mi-Pilo Ophth Sol
- Minims Pilocarpine
- Miocarpine
- Mistura P
- Ocu-Carpine
- Ocusert P 20
- Ocusert Pilo
- Ocusert Pilo-20
- Ocusert Pilo-40
- P.V. Carpine Liquifilm
- Pilagan
- Pilocar
- Pilocar SMP
- Pilocarpal
- Pilocarpin
- Pilocarpine chloride
- Pilocarpine HCl
- Pilocarpine hydrochloride
- Pilocarpine monohydrochloride
- Pilocarpine muriate
- Pilocarpine [Usan:Ban:Jan]
- Pilocarpol
- Pilocel
- Pilokarpin
- Pilokarpin monohydrochloride
- Pilokarpol
- Pilomiotin
- Pilopine HS
- Piloptic-1
- Piloptic-1/2
- Piloptic-2
- Piloptic-3
- Piloptic-4
- Piloptic-6
- Pilostat
- Pilovisc
- Salagen
- Sno Pilo
- Spersacarpine
- Spersacarpine hydrochloride
- Syncarpine
Pilocarpine monohydrochloride Brand Names Mixture
- E-Pilo 1 Ophthalmic Solution (Epinephrine bitartrate + Pilocarpine hydrochloride)
- E-Pilo 2 Ophthalmic Solution (Epinephrine bitartrate + Pilocarpine hydrochloride)
- E-Pilo 4 Ophthalmic Solution (Epinephrine Bitartrate + Pilocarpine Hydrochloride)
- E-Pilo 6 Ophthalmic Solution (Epinephrine Bitartrate + Pilocarpine Hydrochloride)
- E-Pilo-1 Oph Soln (Epinephrine Bitartrate + Pilocarpine Hydrochloride)
- E-Pilo-2 Oph Soln (Epinephrine Bitartrate + Pilocarpine Hydrochloride)
- E-Pilo-4 Oph Soln (Epinephrine Bitartrate + Pilocarpine Hydrochloride)
- E-Pilo-6 Oph Soln (Epinephrine Bitartrate + Pilocarpine Hydrochloride)
- Timpilo 2 (Pilocarpine Hydrochloride + Timolol Maleate)
- Timpilo 4 (Pilocarpine Hydrochloride + Timolol Maleate)
Pilocarpine monohydrochloride Chemical_Formula
C11H16N2O2
Pilocarpine monohydrochloride RX_link
http://www.rxlist.com/cgi/generic3/salagen.htm
Pilocarpine monohydrochloride fda sheet
Pilocarpine monohydrochloride msds (material safety sheet)
Pilocarpine monohydrochloride Synthesis Reference
Preobrashenski et al., Ber. 66, 1187 (1933)
Pilocarpine monohydrochloride Molecular Weight
208.257 g/mol
Pilocarpine monohydrochloride Melting Point
34 oC
Pilocarpine monohydrochloride H2O Solubility
1000 mg/ml
Pilocarpine monohydrochloride State
Solid
Pilocarpine monohydrochloride LogP
1.02
Pilocarpine monohydrochloride Dosage Forms
Drops; Gel; Implant; Liquid; Solution
Pilocarpine monohydrochloride Indication
For the treatment of radiation-induced dry mouth (xerostomia) and symptoms of dry mouth in patients with Sjögrens syndrome.
Pilocarpine monohydrochloride Pharmacology
Pilocarpine is a choline ester miotic and a positively charged quaternary ammonium compound. Pilocarpine, in appropriate dosage, can increase secretion by the exocrine glands. The sweat, salivary, lacrimal, gastric, pancreatic, and intestinal glands and the mucous cells of the respiratory tract may be stimulated. When applied topically to the eye as a single dose it causes miosis, spasm of accommodation, and may cause a transitory rise in intraocular pressure followed by a more persistent fall. Dose-related smooth muscle stimulation of the intestinal tract may cause increased tone, increased motility, spasm, and tenesmus. Bronchial smooth muscle tone may increase. The tone and motility of urinary tract, gallbladder, and biliary duct smooth muscle may be enhanced. Pilocarpine may have paradoxical effects on the cardiovascular system. The expected effect of a muscarinic agonist is vasodepression, but administration of pilocarpine may produce hypertension after a brief episode of hypotension. Bradycardia and tachycardia have both been reported with use of pilocarpine.
Pilocarpine monohydrochloride Absorption
There was a decrease in the rate of absorption of pilocarpine from SALAGEN Tablets when taken with a high fat meal by 12 healthy male volunteers
Pilocarpine monohydrochloride side effects and Toxicity
No information avaliable
Pilocarpine monohydrochloride Patient Information
Patients should be informed that pilocarpine may cause visual disturbances, especially at night, that could impair their ability to drive safely.
If a patient sweats excessively while taking pilocarpine hydrochloride and cannot drink enough liquid, the patient should consult a physician. Dehydration may develop.
Pilocarpine monohydrochloride Organisms Affected
Humans and other mammals